Assuming you have already mastered those let s draw a structure and name it simply based on the molecular formula. In today s post we will talk about the iupac rules of nomenclature for naming alkanes and alkyl halides.
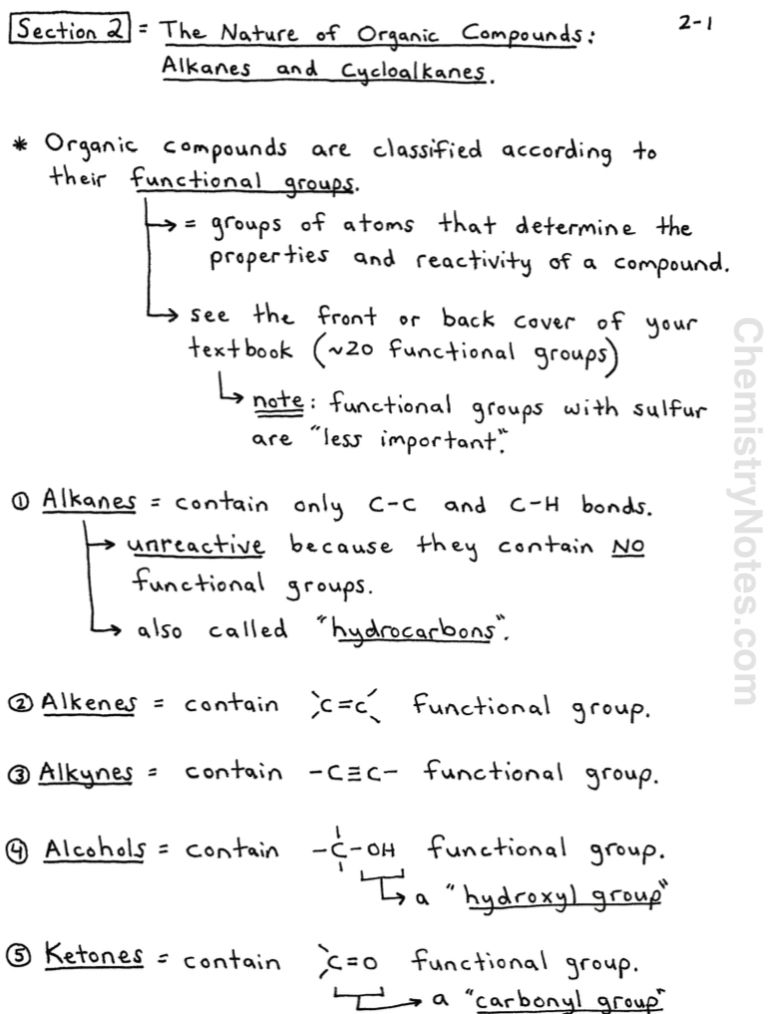
Alkanes And Cycloalkanes Notes Chemistry Notes Functional Groups Organic Chemistry Chemistry Textbook
Determine the parent chain.

Nomenclature worksheet 1 naming alkanes and cycloalkanes. Number the substituents of the ring so that the sum of the numbers is the lowest possible. Numbering generally starts from the end of the chain which is closest to the group named in the suffix 4. Showing top 8 worksheets in the category iupac nomenclature practice.
Use the cycloalkane as the parent chain if it has a greater number of carbons than any alkylsubstituent. Iupac rules for alkane nomenclature 1. The first thing you need to do before learning the iupac rules for systematic nomenclature is making sure you know the names of the first ten alkanes.
However the common names do not generally follow the basic iupac nomenclature rules. Identify and name groups attached to this chain. Number the chain consecutively starting at the end nearest a substituent group.
The general formula of the cycloalkanes is c nh 2n where n is the number of carbons. Designate the location of each substituent group by an appropriate number and name. Some of the worksheets displayed are naming organic compounds practice practice 8 1 give the iupac name of each of the following naming organic compounds bcpldtpbc note 201306 acc j naming hydrocarbons work and key nomenclature in organic chemistry chemistry 1a nomenclature work 1 chemistry 10 organic chemistry.
Find the longest continuous carbon chain. Number the carbons starting closest to the first branch. Following are basic rules for naming cycloalkanes.
Cyclic hydrocarbons have the prefix cyclo. Comprised of just carbon and hydrogen cc h h hh h h cc h hh h hcch saturated no pi bonds unsaturated one or more bonds alkanes alkenes alkynes h h h h h h benzene naming alkanes hc h h h c ch 4 methane c 2h 6 ethane c 3h 8 propane c 4h 10 c 5h 12 c 6h 14 c 7h 16 c 8h 18 c 9h. Alkanes alkane systematic nomenclature cycloalkanes conformations of alkanes conformations of cycloalkanes conformations of alkylcyclohexanes preview you learned in the chapter 1 that all organic molecules have carbon skeletons.
The naming of cycloalkanes follows a simple set of rules that are built upon the same basic steps in naming alkanes. Name the groups attached to the chain using the carbon number as the locator. Iupac rules for nomenclature the naming of substituted cycloalkanes follows the same basic steps used in naming alkanes.
Alkanes and cycloalkanes 1. Find and name the longest continuous carbon chain. If an alkyl chain off the cycloalkane has a greater number of carbons then use the alkyl chain as the parent and the cycloalkane as a cycloalkyl substituent.
Nomenclature of alkane and cycloalkanes each student work group choose a leader reads activity out loud poses questions to group facilitator makes sure everyone is participating equally directs the conversation recorder takes note on discussion learned concepts and any conclusions and presenter announces conclusions or presents activity to class. These carbon skeletons show great diversity in the ways that c atoms bond to each other and in their three.

Nomenclature Of Alkanes Organic Chemistry Readchemistry Com Organic Chemistry Chemistry Structural Formula

Degree Of Substitution Is Also Used In Naming Organic Molecules Organic Molecules Organic Chemistry Study Chemistry

Carboxylic Acids Organic Chemistry Notes In 2020 Organic Chemistry Notes Chemistry Notes Organic Chemistry Cheat Sheet

Naming Alkanes By Iupac Nomenclature Rules Practice Problems Chemistry Steps In 2020 Chemistry Functional Group Methyl Group

Gram Formula Mass Worksheet Chemistry Notes Chemistry Worksheets

Coschemistry Lesson 6 05 Naming Alkanes Chemistry Worksheets Naming Chemical Compounds Worksheet Practices Worksheets

Organic Chemistry 101 Nomenclature Organic Chemistry Study Organic Chemistry Teaching Chemistry

Naming Complex Substituents In Alkane Nomenclature Chemistry Lessons Chemistry Physical Chemistry

Naming Alkenes By Iupac Nomenclature Rules Chemistry Steps In 2020 Organic Chemistry Chemistry Lessons Teaching Chemistry

Naming Alkanes By Iupac Nomenclature Rules Practice Problems Chemistry Steps In 2020 Chemistry Methyl Group Functional Group

Naming Alkanes By Iupac Nomenclature Rules Practice Problems Chemistry Steps In 2020 Chemistry Lessons Chemistry Study Chemistry

E1 Elimination Mechanism Kinetcis Practice Problems Reactions Organic Chemistry Chemistry

Naming Alkanes By Iupac Nomenclature Rules Practice Problems Chemistry Steps In 2020 Chemistry Methyl Group Functional Group

Alkanes Study Chemistry Chemistry Lessons Organic Chemistry Study

Identifying The Parent Chain When Naming An Organic Molecule Chemistry Lessons Chemistry Organic Molecules

Organic Chemistry Notes Alkanes And Cycloalkanes In 2020 Organic Chemistry Chemistry Notes Organic Chemistry Notes

Ions And Ionic Compounds Worksheet Answer Key In 2020 Chemistry Basics Ionic Compound Polyatomic Ion

Alphabetical Priority Of Prefixes Prefixes Chemistry Study Chemistry

Cycloalkane And Cycloalkene Nomenclature Difference Organic Chemistry Study Study Chemistry Organic Chemistry