Manganese 42 25 17 manganese 40 of protons 25 25 of neutrons 17 15 of electrons 25 25 germanium 62 germanium 64 of protons 32 32 of neutrons 30 32 of electrons. Fill in the table with the correct information.
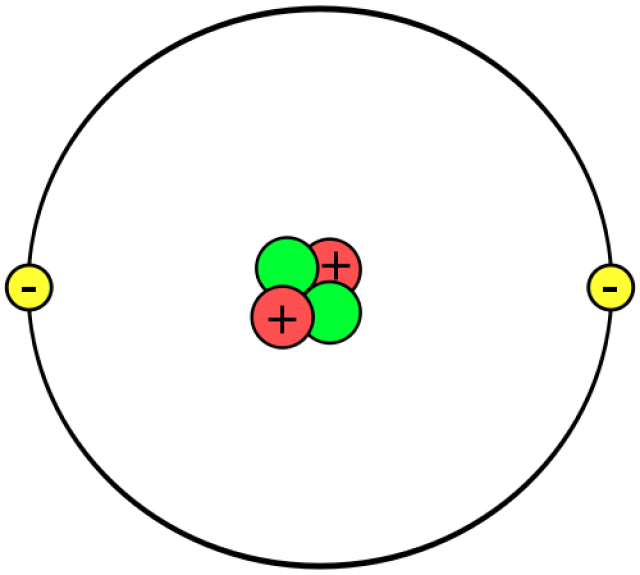
Ways To Make An Atom Model Atom Model Atom Diagram Atom Model Project
Isotopes are all atoms of an element alike.

Isotopes worksheet are all atoms of an element alike. Use your periodic table and the information provided. Isotope isotope notation atomic protons electrons neutrons oxygen 16 8 8 8 8 bromine 80 35 35 35 45 uranium 235 92 92 92 143 copper 64. A do all isotopes of an element have the same atomic number.
Combined together in. This is just a sampling of what we will address. View isotopes docx from science 412 at baldwin senior high school baldwin.
Give at least one example or counter example from model 1 that supports your answer. Mass of isotope abundance 36 96590 24 47 34 96885 75 53 atoms and isotopes worksheet. Considering your answers to question 11 work with your group to write a definition of isotope using a sentence.
The following activity will help you learn the important. How does the combination of subatomic particles affect the mass and charge of an atom. No carbon 12 and carbon 13 are isotopes of carbon but have different mass numbers.
The following activity will help you learn the important structural characteristics of an atom. Isotopes or different elements showing top 8 worksheets in the category isotopes or different elements. Assume all atoms are neutral.
All atoms of one element are. Compounds consist of atoms of two or more atoms. The particles that make up elements are called.
Isotopes student worksheet 1 doc isotopes are all atoms alike why the following activity will help you learn the important structural isotopes student worksheet 1 doc isotopes are all atoms. In his atomic theory dalton proposed that atoms were the smallest particle capable of existence. Fill in the isotope names and any missing information on the chart.
The same but they differ from the atoms of all other. Yes all the magnesium atoms have 12 protons so they all have an atomic number of 12. Remind students that all atoms are isotopes.
B do all isotopes of an element have the same mass number. Naturally occurring chemical elements are usually mixtures of isotopes and so their atomic masses are weighted averages of the masses of the isotopes in the mixture. Some of the worksheets displayed are isotopes he sai isotopes atoms and ions isotopes are all atoms of an element alike work lesson plan understanding isotopes bag o isotopes.
Isotopes are atoms of the same element with different mass numbers. Give at least one example or counter. How do wc classify atoms.
Distribute the science netlinks lab packet isotopes of pennies to each student. Isotopes are all atoms of an element alike.
Https Www Conejousd Org Portals 49 Departments Science Cord 202015 Ch 204 20and 2025 20atom 20and 20nuclear H 20hw 20and 20worksheets 20key Pdf Ver 2017 09 26 162008 360

See The Electron Configuration Diagrams For Atoms Of The Elements Electron Configuration Atom Physics

Atoms And Molecules Worksheet Luxury 38 Best Images About Atoms Molecules On Pinterest In 2020 Chemistry Lessons Science Lessons Teaching Chemistry

Accomplishedgeneral Atoms And Ions Worksheet Atomandhispackage Atomandmarkdown Atomsandmanmech Atomsbandinkontinenz Atomsbandurologie

Iv Isotopes 2 Or More Atoms Of The Same Element Having The Same Number Of Protons But Different Numbers Of Neutrons Atomic Theory Chemistry Lessons Protons

Isotopes Atoms With The Same Number Of Protons But Different Numbers Of Neutrons Ex Carbon 12 Vs Carbon 14 These Ato What Is Atom Atomic Structure Atom

Isotopes And Atomic Mass Teaching Chemistry Secondary Science Classroom Physical Science

Atoms Ions Isotopes Wmv Improve Your English Physical Science Atom

Isotopes And Atomic Mass Chemical Science Atom Teaching Chemistry

The Atom Chemistry Is My Jam Atom Electron Configuration Chemistry Help

Chemistry How To Calculate Atomic Mass Of An Element Isotopes Isotope Notation Atomic Mass Unit Relative Atomic Mass Chemistry Lessons Teaching Chemistry

Atoms And Isotopes Worksheet Answers Lovely Atoms And Isotopes Worksheet In 2020 Worksheets Practices Worksheets Answers

Making Models Of Atoms And Isotopes 7th 9th Grade Lesson Plan Biology Worksheet Chemistry Worksheets Lesson Planet

Protons Neutrons And Electrons Worksheet Subatomic Structure Worksheet Chemconnections In 2020 Chemistry Worksheets Word Problem Worksheets Fraction Word Problems

Why Atomic Weight And Atomic Mass Are Not The Same Thing In 2020 Relative Atomic Mass Atom Earth Gravity

Pin By Alison Roughsedge On Chemistry Education Chemistry Education Atom Teaching Chemistry
Https Www Livingston Org Cms Lib4 Nj01000562 Centricity Domain 826 Pogil 20isotopes 20answer 20key Pdf

Difference Between Isotope And Radioisotope Definition Properties Examples Chemistry Lessons Electron Configuration Definitions

Pin By Aguerra On Chemistry Chemistry Wholeness Protons

