As the number of neutrons increase in the isotope the nuclear charge a increases b decreases c remains the same. Complete the following table.
Nuclear Reaction Worksheet Answers In 2020 Nuclear Reaction Chemistry Worksheets Worksheets
The chapter on atoms molecules and ions introduced the basic idea of nuclear structure that the nucleus of an atom is composed of protons and with the exception of ce 1 1h neutrons.

Isotopes and nuclear chemistry worksheet. Carbon is composed primarily of two isotopes. Cobalt 60 is produced when a radioactive isotope undergoes beta decay. An anion with a charge of 1 containing 53 protons and 74 neutrons 2.
Nuclear chemistry is the study of reactions that involve changes in nuclear structure. Nuclear chemistry practice worksheet key exercise 1. Chemistry chemistry quizzes.
Isotopes and nuclear chemistry worksheet chemistry 2a name section 1. Atoms of a given element which have the same number of protons but different numbers of neutrons are called isotopes. Uranium 234 is produced when a radioactive isotope undergoes alpha decay.
For example the atomic mass of carbon is reported as 12 011 amu. 13 an atom with 29 protons and 36 neutrons is an isotope of a si b cu c kr d cl 14 carbon exists as three naturally occurring isotopes. The proper way to notate an isotope is to use the element s name or symbol with a dash and the element s mass number.
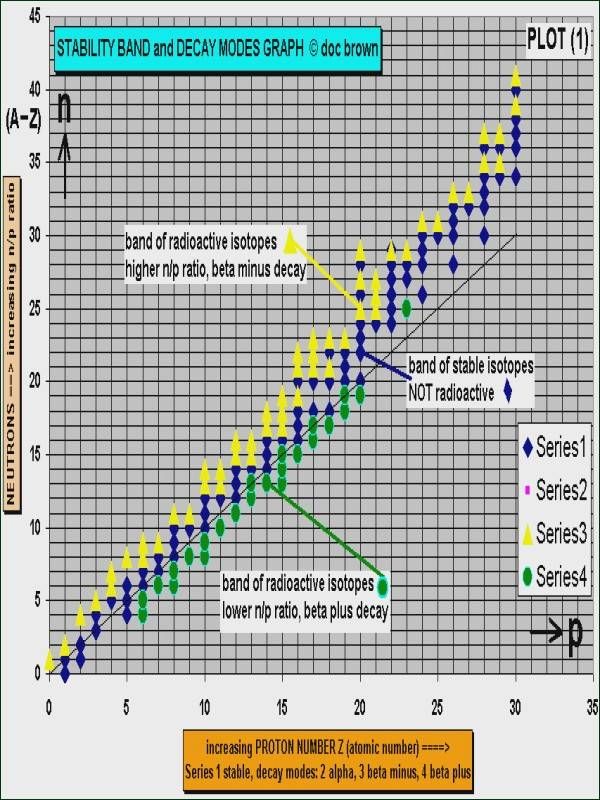
Containment on site storage and off site disposal remember that every radioactive substance has a half life some only a few months others hundreds of thousands of years. Write the atomic symbols for each isotope mass number two isotopes of gallium one having 37 neutrons and the other having 39 neutrons. A nuclear nomenclature can be adopted to describe an specific nucleus that explicitly indicates both a and z.
Nuclear chemistry an introduction. Write complete nuclear equations for the following processes. Argon 40 is bombarded with a proton.
Abundance of isotopes h worksheet 4 3 keg 20m name the atomic mass for each element is reported on the periodic table. U 235 u 236 and u 238 refer to three isotopes of uranium with mass numbers of 235 236 and 238. Isotope mass number of protons of neutrons of electrons strontium 90 90 38 52 38 222rn 222 86136 exercise 1.
Types of nuclear waste spent fuel rods dismantled nuclear power plants military radioisotopes used in many hospitals 2. Identify the unknown element in each to complete the following nuclear equations. The simplest example of an atom with different isotopes is hydrogen.
Isotopes are nearly identical and have the same number of protons e g. Thus isotopes have the same position in the periodic table the same chemical properties and the same atomic charge. Charger applicable x element symbol see periodic table atomic number 2 a cation with a charge of 3 containing 13 protons and 14 neutrons.
The inhalation of radon 222 and its decay to form other isotopes poses a health hazard. For the following quiz you will need a periodic table. This number is a weighted average of the masses of each of the isotopes of an element.
Select the best answer to the question. C 12 c 13 and c 14. Gcc chm152ll nuclear chemistry summer practice worksheet p 3 of 4 exercise 2.

Nuclear Chemistry Review Worksheet Nuclear Fission And Fusion In 2020 Gcse Science Chemistry Worksheets Chemistry Review

Atomic Structure And Nuclear Chemistry Homework Unit Bundle Chemistry Chemistry Worksheets Atomic Structure

Nuclear Chemistry Worksheet Doc Chemistry Worksheets Math Review Worksheets Chemistry

21 3 Radioactive Decay Chemistry Nuclear Medicine Chemistry Review Chemistry Worksheets

Atomic Structure And Nuclear Chemistry Homework Pages Atomic Structure Chemistry Worksheets High School Chemistry

Atomic Structure Worksheet Science Education Teaching Chemistry Chemistry Classroom Chemistry Worksheets

Atomic Structure Worksheet Chemistry Worksheets Teaching Chemistry Chemistry Classroom

Chemistry How To Calculate Atomic Mass Of An Element Isotopes Isotope Notation Atomic Mass Unit Relative Atomic Mass Chemistry Lessons Teaching Chemistry

Atomic Structure And Nuclear Chemistry Homework Pages Chemistry Chemistry Worksheets Atomic Structure

Atomic Structure And Nuclear Chemistry Homework Unit Bundle High School Chemistry Chemistry Classroom Chemistry Worksheets

Nuclear Fission And Fusion Reactions Cornell Doodle Notes Doodle Notes Nuclear Physics Notes

Atomic Structure And Nuclear Chemistry Homework Unit Bundle Atomic Structure Nuclear Chemistry Classroom High School Chemistry Teaching Chemistry

Atoms Worksheet Teaching Chemistry Chemistry Classroom Chemistry Lessons

Protons Neutrons And Electrons Worksheet Subatomic Structure Worksheet Chemconnections In 2020 Chemistry Worksheets Word Problem Worksheets Fraction Word Problems

Worksheet That Covers Radioactive Decay Half Life Differentiating Synthetic And Naturally Occurring Elements And Naming New Elements Science Half Life Life

Structure Of An Atom Doodle Notes Middle School Chemistry Middle School Chemistry Teaching Chemistry Teaching Science

Pin On Super Teacher Worksheets

Balancing Nuclear Equations Worksheet Answers Nuclear Decay Worksheet Answers Key Beautiful Tom Schoderbek In 2020 Chemistry Worksheets Worksheets Chemistry

Atomic Structure Worksheet Chemistry Worksheets Teaching Chemistry Chemistry Classroom

