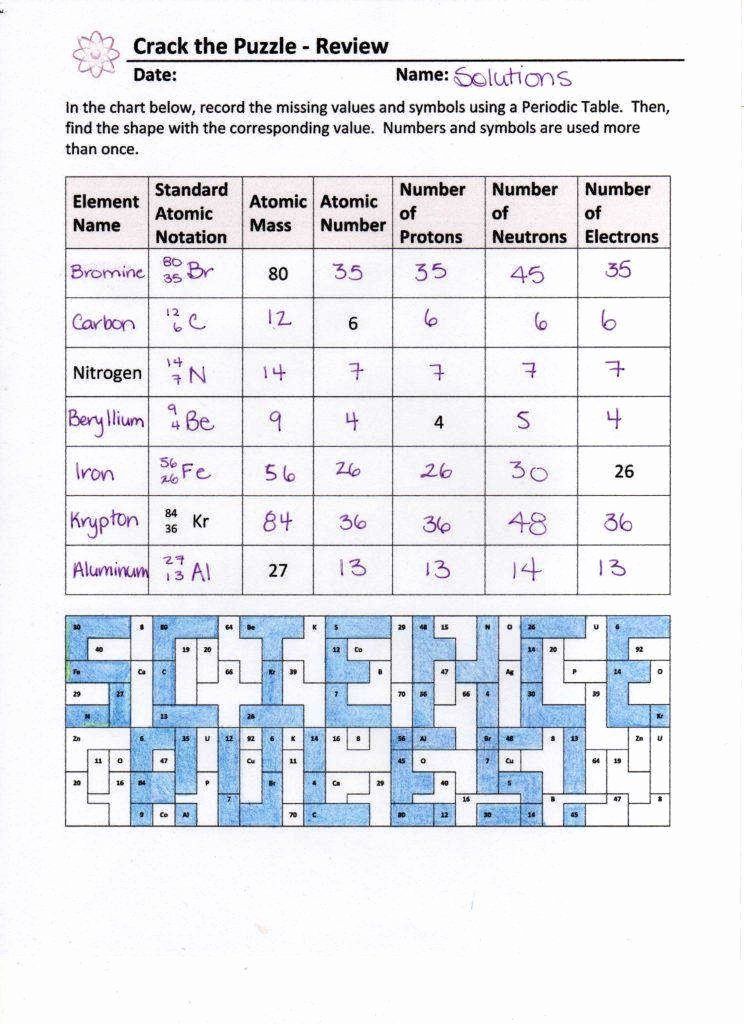
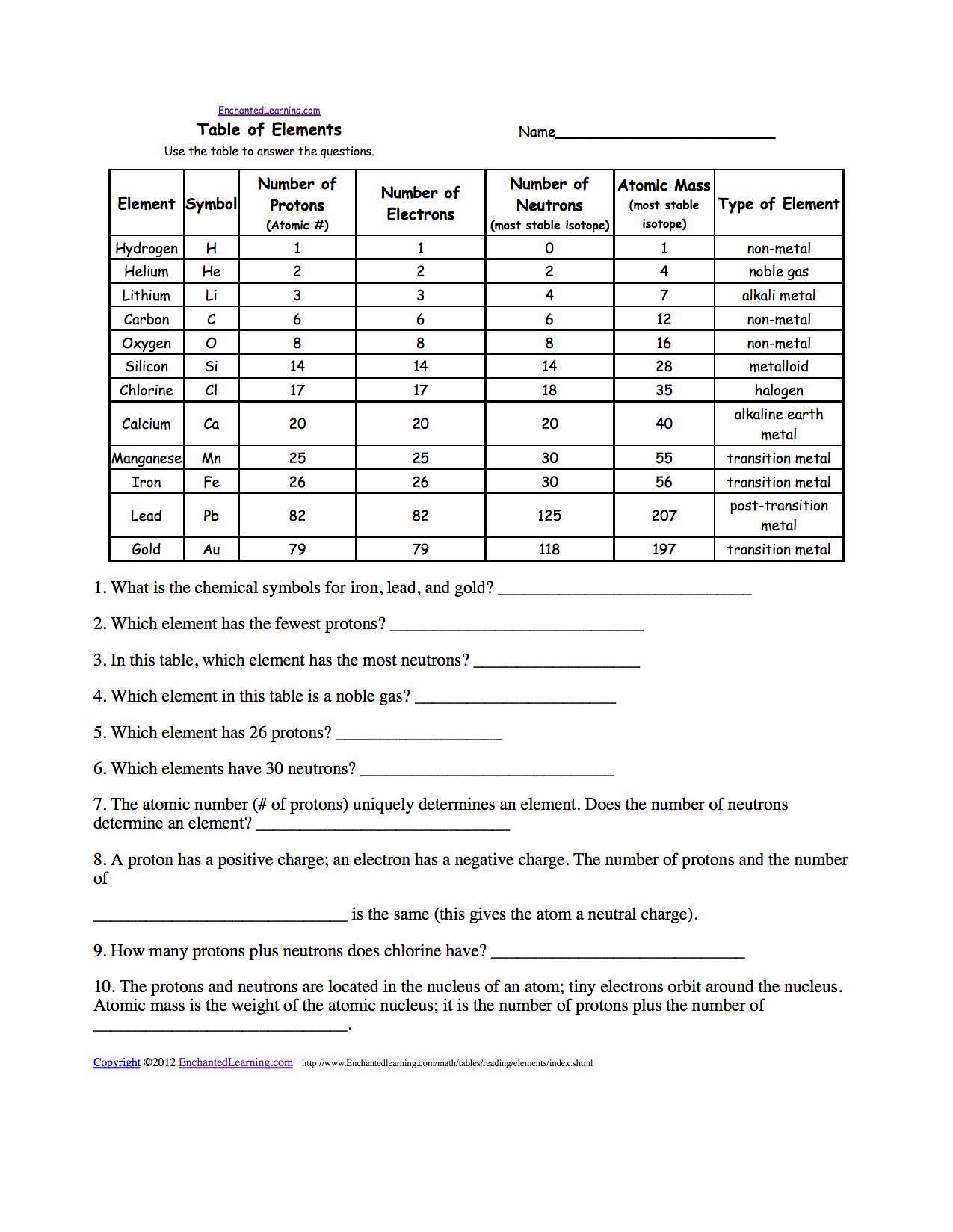
Element name symbol atomic number number of electrons number of neutrons number of protons mass number atomic number atomic mass. Chemistry q a library variations in average atomic mass may be observed for elements obtained from different sources.
Isotope Practice Worksheet Answers Beautiful Isotopes Worksheet In 2020 Physical Science Chemistry Review Chemistry Lessons
The number indicates the isotope s mass number.
Chemistry isotopes worksheet answers. êsym name 24 mg 30 108pd 13 35 s2 112 cd2 52 2 24 of atom or atomic mass of 53 94 56 no 30 125 69 80 ion. Isotope practice worksheet name. No carbon 12 and carbon 13 are isotopes of carbon but have different mass numbers.
Use isotope notation to determine. Calculate its average atomic mass. View isotopes worksheet 1 key pdf from chemistry 1305 at university of texas el paso.
Recall that a mass number z atomic number and ions have charge. They have the same number of protons and electrons as the element but different mass numbers and number of neutrons. Isotope mass both varies in.
The natural abundance for boron isotopes is 19 9 10b and 80 1 11b. Model 1 that supports your answer. Do all isotopes of an element have the same mass number.
88 94 chapter 3 in your chemistry book click on the link to go directly to the web version of chapter 3. Here are three isotopes of an element. List the number of protons and neutrons for each.
Considering your answers to question 1 1 write a definition of isotope using a grammatically correct sentence. 3 so fijpes w 12. Rubidium is a soft silvery white metal that has two common isotopes 85rb and 87rb.
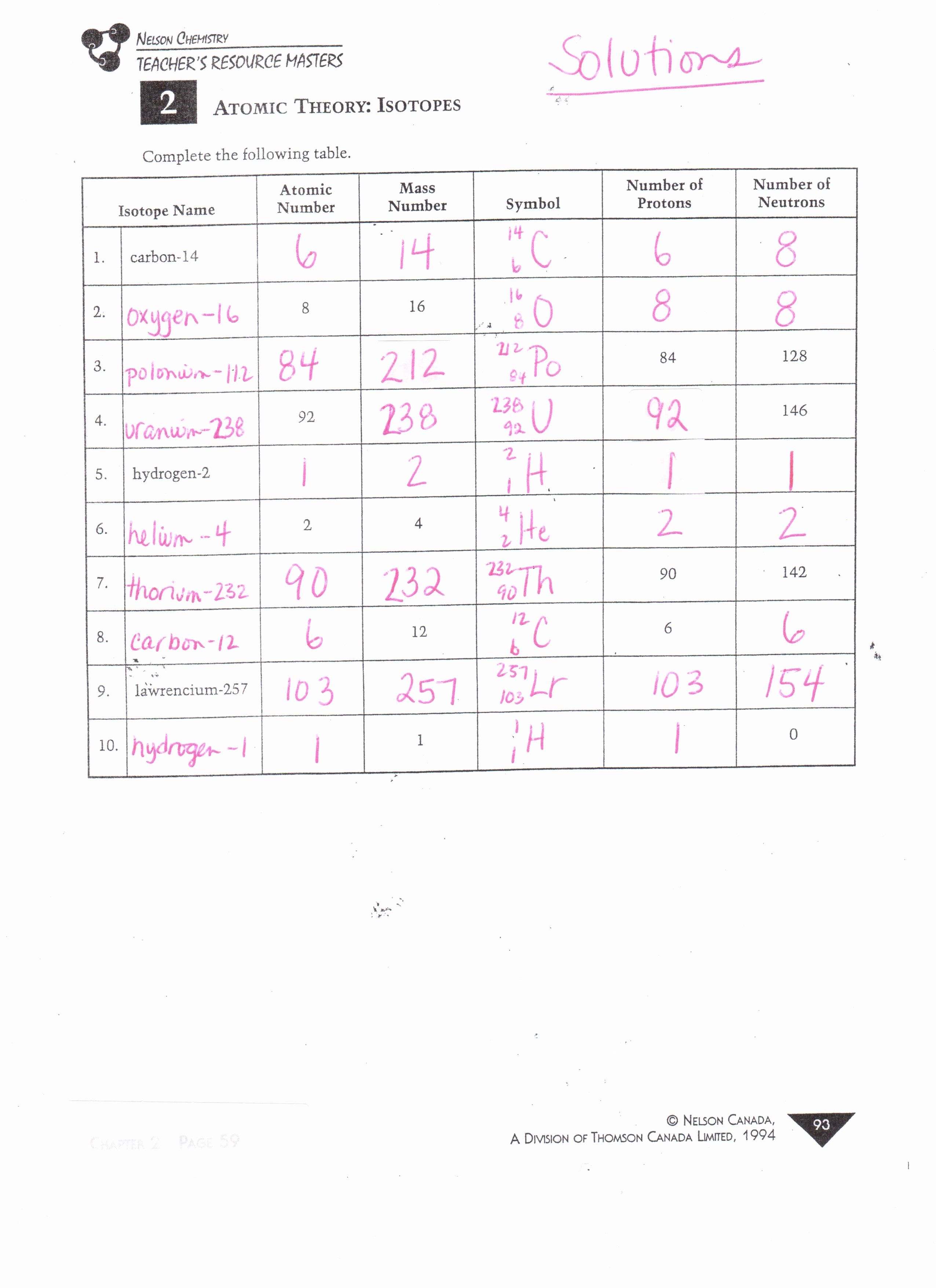
Atomic structure and isotopes answers give the chemical symbol for the following isotopes including the atomic number and mass number. Isotopes are atoms of the same element with different mass numbers. S sax ne h d ç.
What is an isotope. Give at least one example or counter example from model 1 that supports your answer. Isotopes are versions of the same element.
Your group must come to consensus on this definition. Calculate boron s atomic mass. Considering your answers to question 11 work with your group to write a definition of isotope using a sentence.
When chlorine occurs in nature there are three atoms of chlorine 35 for every one atom of chlorine 37. Hydrogen is 99 1h 0 8 2h and 0 2 3h. Atoms and isotopes worksheet answers worksheet center.
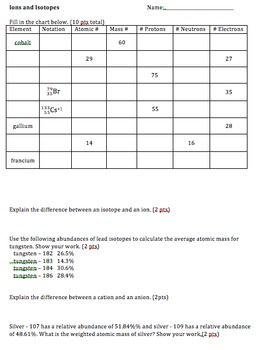
Element symbol atomic number mass number isotopes lead pb 82 207 3 206 207 208 gold au 79 197 197 krypton k 36 83 8 80 82 83 84 86 iron fe 26 55 847 54 55 57 hydrogen h 1 1 008 1 2 3. Use the information given in the following table to answer the questions. What does the number next to isotopes signify.
Metal non metal or metalloid. 6 12c 14 6 13c 6 c a. Answer the questions based on the above reading.
Chemistry worksheet isotope notation name. Atom atom atom atom atom atom number number 10 56 25 85. Atomic theory and structure.

Isotope Practice Worksheet Answer Key Lovely Unit 2 Chapters 4 5 6 Mrs Gingras Chemistry Page In 2020 Chemistry Worksheets Fractions Worksheets Worksheet Template

Atoms And Isotopes Worksheet Answers Inspirational Chemistry Worksheets 2 For Interactive Noteboo In 2020 Chemistry Worksheets Teaching Chemistry Interactive Notebooks

Isotopes Review A Chemistry Coloring Page Editable Chemistry Homeschool Science Science Chemistry

Isotopes Worksheet Jpg 357 462 Physical Science Science Worksheets Practices Worksheets

Contains Atomic Number Worksheet Mass Number Worksheet Atomic Mass Worksheet And Bohr Model Worksheet Worksheets In 2020 Chemistry Worksheets Chemistry Worksheets

Isotopes Ions And Atoms Worksheet Fresh Solved Atomic Structure And Chemical Nomenclature Workshe In 2020 Chemistry Worksheets Atomic Structure Worksheets

Atomic Structure Worksheet And Periodic Table Chemistry Worksheets Atomic Structure Chemistry Classroom

Isotope Practice Worksheet Answer Key Lovely 12 Best Of Protons Neutrons Electrons Practice In 2020 Practices Worksheets Homework Worksheets Chemistry Worksheets

Pin On Super Teacher Worksheets

Making Models Of Atoms And Isotopes 7th 9th Grade Lesson Plan Biology Worksheet Chemistry Worksheets Multi Step Equations Worksheets
Ions And Isotopes Practice Two Versions Practices Worksheets Aufbau Principle Biology Lesson Plans
Isotope Practice Worksheet Answer Key Beautiful Worksheet Element Symbols Worksheet Grass Fed In 2020 Practices Worksheets Chemistry Worksheets Word Problem Worksheets

Pin On Customize Design Worksheet Online

Pin By Taylor Wene On Chemistry Practices Worksheets Worksheets Biology Lesson Plans
Ions And Isotopes Worksheet Google Search Chemistry Lessons Practices Worksheets Chemistry Worksheets

Atoms Isotopes And Molecules Activity Sheet With Answer Key Tpt Science Lesson Plans Answer Keys Science Lessons

Isotope Practice Worksheet Answer Key Beautiful Parative Anatomy Worksheet Free Printable Worksheets In 2020 Practices Worksheets Worksheets Biology Lesson Plans

Chem Unit 2 Worksheet Bundle Atoms Ions Isotopes Compo Chemistry Worksheets Interactive Notebooks Teaching Chemistry

Atoms And Isotopes Worksheet Answers Lovely Isotopes And Atomic Mass Worksheet Answer Key In 2020 Chemistry Worksheets Spelling Worksheets Worksheets