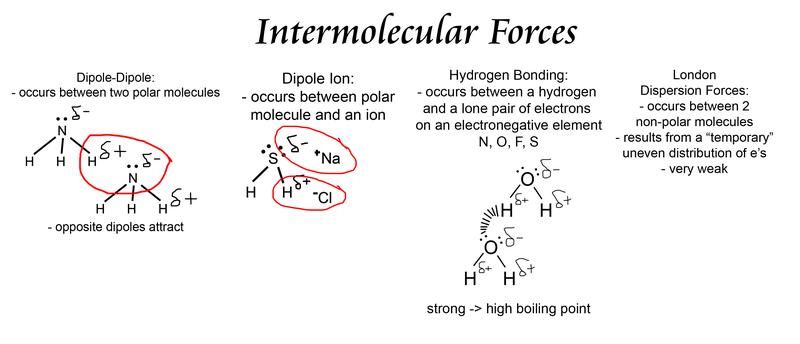
This is the strongest intermolecular force occurs in molecules with h f h o and h n bonds. Positive charge on hydrogen is attracted to unshared.
Msextavourscienceclasses Intermolecular Forces Nastas Lena Intermolecular Force Ap Chem Chemistry
Rank the ionic bond strength for the following ionic formulas 1 being strongest.

Chemistry intermolecular forces worksheet answers. If the molecule is polar but doesn t have the bonds mentioned above its primary intermolecular force is a dipole dipole force. Intermolecular forces the tendency of a substance to be found in one state or the other under certain conditions is largely a result of the forces of attraction that exist between the particles comprising it. What is the strongest intermolecular force present for each of the following molecules.
Has only very weak london dispersion forces lowest b p. Intermolecular forces imf can be ranked qualitatively using coulomb s law. Solubility boiling points melting points.
Intermolecular forces worksheet answers are on page 3 4. Intermolecular forces worksheet answers 1 using your knowledge of molecular structure identify the main intermolecular force in the following compounds. Stronger intermolecular forces cause higher melting and boiling points.
Intermolecular forces wkst answers due no due date points 0. This assignment was locked may 2 2016 at 8am. You may find it useful to draw lewis structures to find your answer.
Force of attraction between the positive end of one molecule and the negative end of another. If the molecule is nonpolar then the primary intermolecular force is a london dispersion force. If the molecule has an h f h o or h n bond anywhere on it the primary intermolecular force is hydrogen bonding.
Don t post outcomes results to learning mastery. Remove points from rubric. Worksheet intermolecular forces chemical bonds are intramolecular forces which hold atoms together as molecules.
Al 2s 3 mgo mgcl 2 nacl strategy. General chemistry ii jasperse intermolecular forces ionic bond strength phase diagrams heating curves. Intermolecular forces worksheet answers pdf i ll write free form comments when assessing students.
Predict the molecular shape of each of the following. Do the problems on your own before looking at the answers. Effects of intermolecular forces.
1 hydrogen h 2 london dispersion forces 2 carbon monoxide co london dispersion forces 3 silicon tetrafluoride sif 4 london dispersion forces 4 nitrogen tribromide nbr 3 dipole dipole forces 5 water h 2 o hydrogen bonding 6 acetone ch 2. List all types of imfs that would occur in each of the following you should have a good. Intermolecular forces exist between atoms and molecules determining their physical properties.
Available apr 29 2016 at 3pm may 2 2016 at 8am 3 days. Extra practice problems 1. We will concentrate on the forces between molecules in molecular substances which are called intermolecular forces.
The strength of intermolecular forces present in a substance is related to the boiling point and melting point of the substance.

Ap Chemistry Core Concept Cheat Sheet 12 Atomic Structures Key Chemistry Terms Boxes And Arrow Chemistry Worksheets College Chemistry Chemistry Education

505 Stmatterimf Htm Chemistry Education Intermolecular Force Chemistry Lessons

Intermolecular Forces Worksheet Mazes 3 Levels Distance Learning In 2020 Intermolecular Force Force Ap Chemistry

Van Der Waals Intermolecular Forces Google Search Intermolecular Force High School Chemistry Mcat Study

Imfa Summary Table Intermolecular Force Chemistry Lessons Chemistry

Intermolecular Forces Worksheet High School Chem 112 Intermolecular Forces Chang F In 2020 Math Fractions Worksheets Kids Worksheets Printables Word Problem Worksheets

Pin On Worksheet Templates For Student

Gram Formula Mass Worksheet Chemistry Notes Chemistry Worksheets

Pin By Megan Cozort On Chem In 2020 Intermolecular Force Hydrogen Bond Chemistry

Intermolecular Forces Worksheet High School Grade 12 Organic Chemistry Notes In 2020 Chemistry Worksheets Word Problem Worksheets Math Fractions Worksheets

Vsepr Theory 3 Levels Of Mazes Vsepr Theory Kids Worksheets Printables Radical Expressions

Intermolecular And Intramolecular Forces Mr Lowe S Science And Chemistry Chemistry Education Chemistry Teaching Chemistry

Chemistry Formula Mass Worksheet F Jram Formula Mass Nome W Defermine Rhe Groum Chemistry Worksheets Chemistry Lesson Planet

Chemistry Notes Liquids Solids And Intermolecular Forces In 2020 Chemistry Notes Intermolecular Force Chemistry

Intermolecular Forces Cmap Intermolecular Force Force Physics Chemistry

Learn Quiz On Kinetic Interpretation Of Temperature Chemistry Quiz 262 To Practice Free Chemistry Mcqs Que College Chemistry Chemistry This Or That Questions

Chart Of Intermolecular Forces For Ap Chemistry Students Ap Chemistry Teaching Chemistry Chemistry Lessons

Intermolecular Forces And Hydrogen Bonding Review Question Trail Intermolecular Force This Or That Questions Hydrogen Bond

Chemistry Puzzle Color By Number Intermolecular Forces Intermolecular Force Chemistry Chemistry Lessons

