Therefore boron 11 is more abundant because the mass number is closer to the atomic mass. Boron exists in two isotopes boron 10 and boron 11.
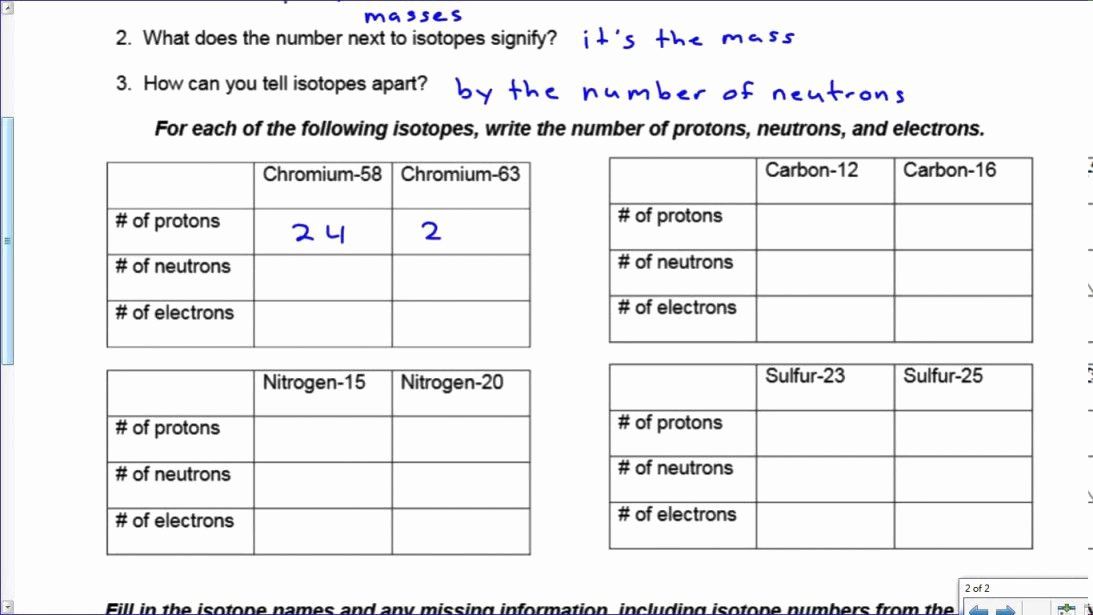
Isotope Practice Worksheet Answer Key New Ions And Isotopes Worksheet In 2020 Practices Worksheets Persuasive Writing Prompts Matter Worksheets
Most atoms have isotopes that happen naturally.

Atomic structure isotopes worksheet answers. Which two subatomic particles are located in the nucleus of an atom. Atomic structure and isotopes answers give the chemical symbol for the following isotopes including the atomic number and mass number. I the 13c isotope has a relative isotopic mass of 13 00.
Define the term relative isotopic mass. Do all isotopes of an element havc the same atomic number. Based on the atomic mass which isotope should be more abundant.
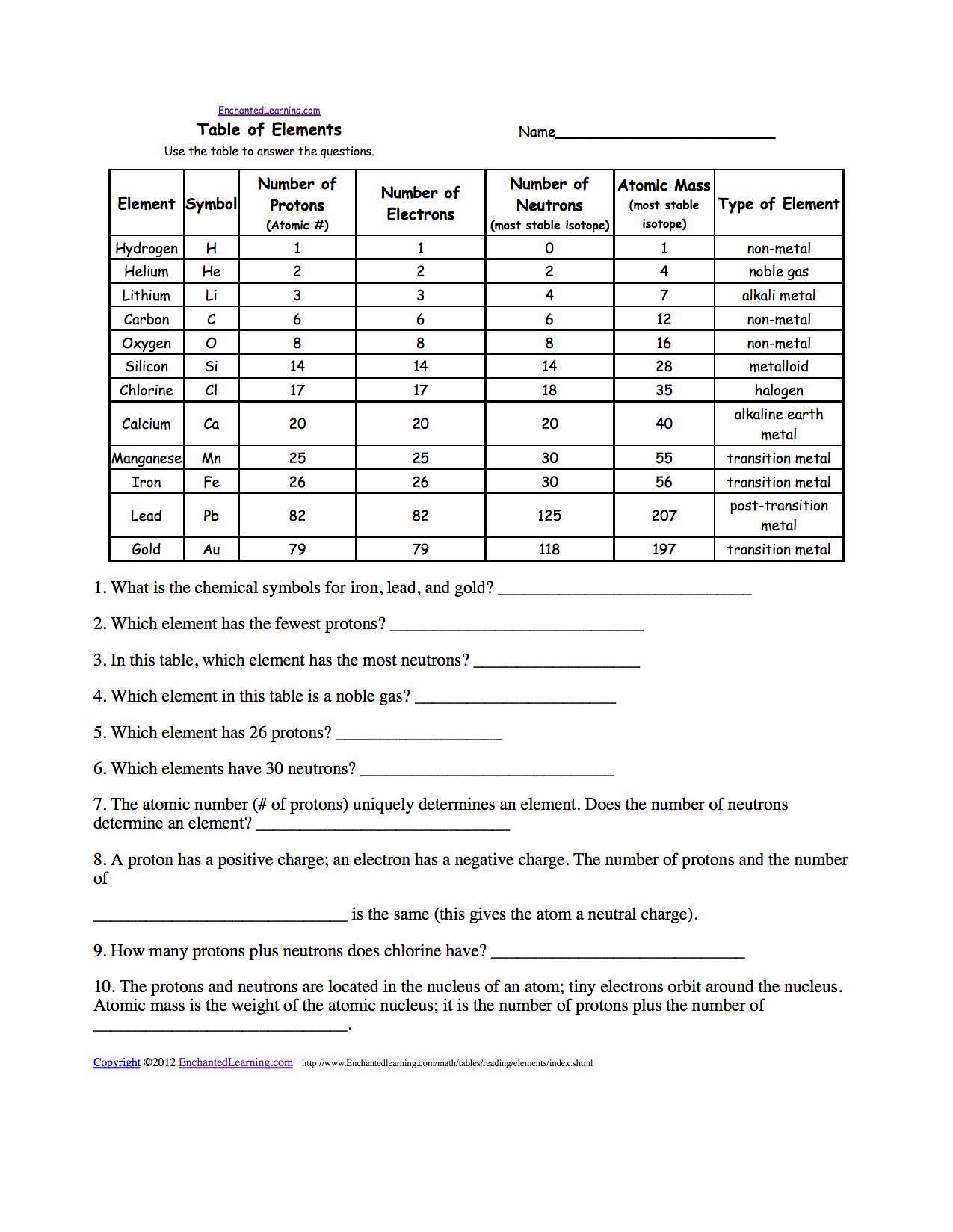
88 94 chapter 3 in your chemistry book click on the link to go directly to the web version of chapter 3. Free chapter 4 atomic structure worksheet answer key consequently simple here. Isotope isotope notation atomic protons electrons neutrons oxygen 16 8 8 8 8 bromine 80 35 35 35 45 uranium 235 92 92 92 143 copper 64.
List the number of protons and neutrons for each. Atomic structure worksheet what type of charge does a proton have. The atomic mass of boron is 10 811.
Lithium 6 is 4 abundant and lithium 7 is 96 abundant. In advance of speaking about atomic structure practice worksheet answers remember to understand that education will be all of our answer to a much better next week as well as learning doesn t just avoid when the school bell rings that will currently being said we give you a number of very simple however educational articles or blog posts and web themes built made for any kind of. Isotope symbol atomic number mass number number of protons number of neutrons 11.
Atomic structure and isotopes answers give the chemical symbol for the following isotopes including the atomic number and mass number. Since atoms are neutral the variety of electrons in an atom is equivalent to the range of protons. What type of charge does a neutron have.
What is the charge of an ion. Later this chapter 4 atomic structure worksheet answers tends to be the scrap book that page 9 30. Give at least one example or counter example from model 1 that supports your answer.
A complete the table below for the atomic structure of the isotopes 12c and 13c. Consider the examples in model 1. Fill in the table with the correct information.
Isotope protons neutrons electrons 12c 6 6 6 13c 6 7 6 2 b a sample of carbon was found to contain 95 of 12c and 5 of 13c. Ho 16 31 15 a. Read free chapter 4 atomic structure worksheet answers he sai 81w 36kr sldu 37kd atomic number mass number protons electrons neutrons isotope name partb.
Atomic structure and isotopes in the atomic structure simulation you will get the opportunity to decide what the. Explain your answer fully or 10 ses. List the number of protons and neutrons for each.
What is the charge of an atom. Mass of isotope abundance 36 96590 24 47 34 96885 75 53 atoms and isotopes worksheet. What type of charge does an electron have.

Atomic Structure Worksheet And Periodic Table Chemistry Worksheets Atomic Structure Chemistry Classroom
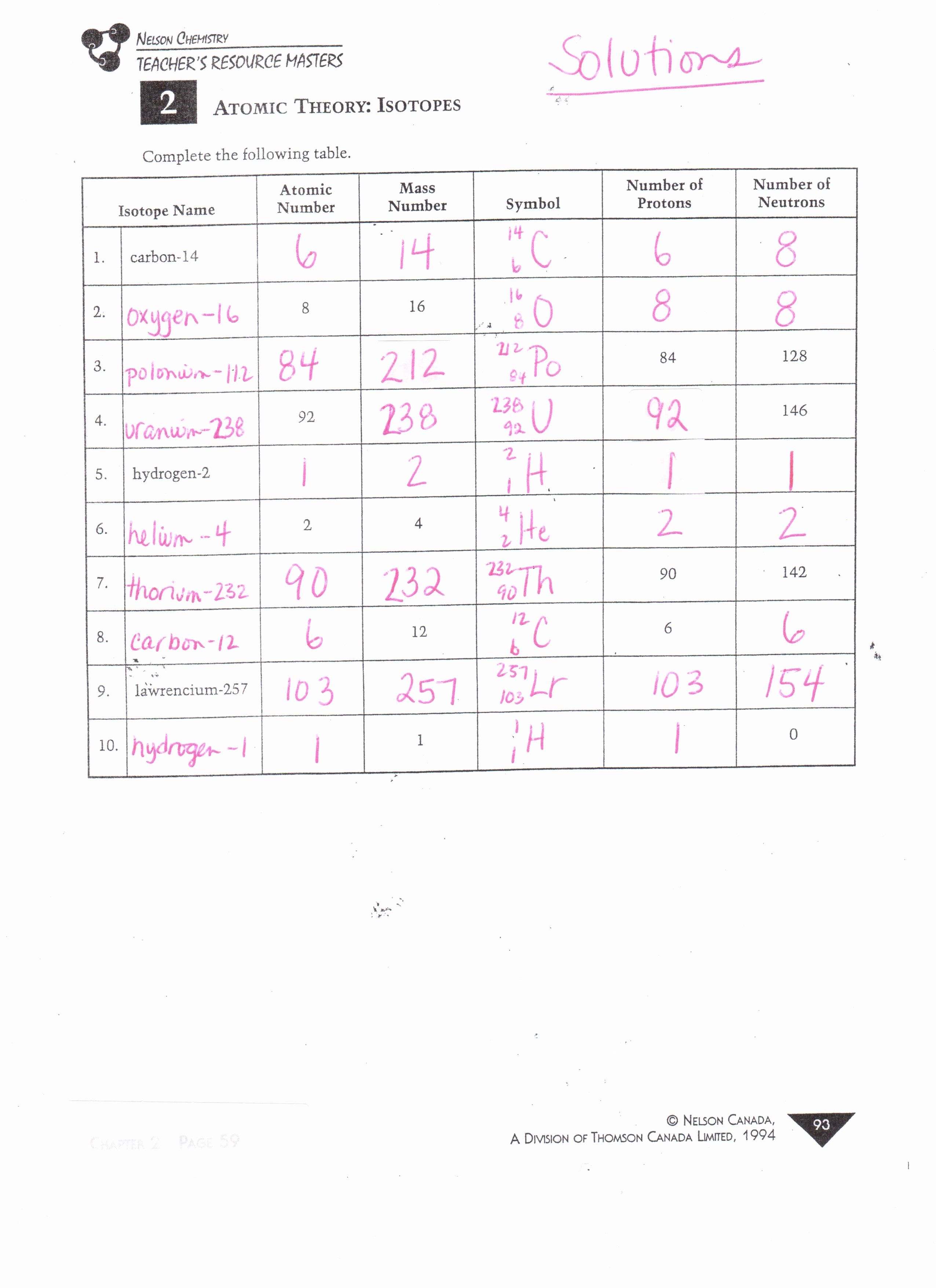
Isotope Practice Worksheet Answer Key Lovely Unit 2 Chapters 4 5 6 Mrs Gingras Chemistry Page In 2020 Chemistry Worksheets Fractions Worksheets Worksheet Template

Isotope Practice Worksheet Answer Key Beautiful Parative Anatomy Worksheet Free Printable Worksheets In 2020 Practices Worksheets Worksheets Biology Lesson Plans

Calculating Parts Of An Atom Practice Worksheet 3 Practices Worksheets Worksheets Atom

Pin On Super Teacher Worksheets

Isotope Practice Worksheet Answer Key Lovely 12 Best Of Protons Neutrons Electrons Practice In 2020 Practices Worksheets Homework Worksheets Chemistry Worksheets

Isotopes Ions And Atoms Worksheet Fresh Solved Atomic Structure And Chemical Nomenclature Workshe In 2020 Chemistry Worksheets Atomic Structure Worksheets

Isotope Practice Worksheet Answer Key Awesome Isotope Practice Worksheet Answer Key Free Prin In 2020 Practices Worksheets Persuasive Writing Prompts Matter Worksheets

Atomic Structure Worksheet Chemistry Worksheets Teaching Chemistry Chemistry Classroom
Ions And Isotopes Worksheet Google Search Chemistry Lessons Practices Worksheets Chemistry Worksheets

Isotope Practice Worksheet Answer Key Unique Learn Isotope Notation Periodic Table Worksheet In 2020 Persuasive Writing Prompts Practices Worksheets Worksheets

Atomic Structure Worksheet Teaching Chemistry Chemistry Classroom Chemistry Worksheets
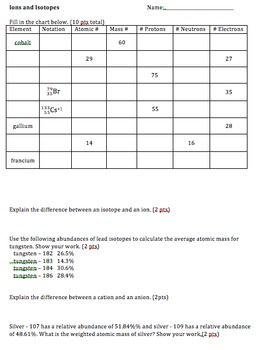
Ions And Isotopes Practice Two Versions Practices Worksheets Aufbau Principle Biology Lesson Plans
Isotope Practice Worksheet Answer Key Beautiful Worksheet Element Symbols Worksheet Grass Fed In 2020 Practices Worksheets Chemistry Worksheets Word Problem Worksheets

Atomic Structure Quiz In 2020 Chemistry Classroom Teaching Chemistry Physical Science

Atomic Structure Worksheet Chemistry Worksheets Teaching Chemistry Chemistry Classroom

Isotopes Worksheet Jpg 357 462 Physical Science Science Worksheets Chemistry Worksheets

Printables Atomic Structure Worksheet Gozoneguide Thousands Of Chemistry Worksheets Atomic Structure Atomic Theory


