Net ionic equation worksheet answers write balanced molecular ionic and net ionic equations nie for each of the following reactions. He then gives you a short introduction to balancing equations and uses the phet site to practice this skill.

Lesson Plan Net Ionic Equations Equations Chemistry Worksheets Teaching Chemistry
David nelson created date.

Ap chemistry net ionic equation worksheet. Net ionic equations for acid base reactions write net ionic equations for each reaction and identify the spectator ions. This type of reaction is called a precipitation reaction and the solid produced in the reaction is known as the precipitate. Honors chemistry name period net ionic equation worksheet.
David nelson last modified by. Assume all reactions occur in aqueous solution. Write a balanced net ionic equation for the overall chemical reaction occurring in the cell.
You can predict whether a precipitate will form using a list of. What is the cell potential ecell. Molecular ion net ionic equations in this video paul andersen shows you how to write balanced equations that describe chemical changes.
A nitrous acid and sodium hydroxide. A set of solubility rules are given at the end of this. The net ionic equation for any weak acid strong base reaction is ha aq oh aq a aq h 2o weak acid hydroxide conjugate base water practice.
Lo mapped the frq s that are listed on a worksheet marked as lo mapped are mapped to specific lo s in the may 2019 ced. Ap topic 12 test a. It should be noted that none of the questions prior to 2020 were actually mapped by the college board to the may 2019 ced since they were all written prior to that document being produced.
Practice problems on net ionic equations page 1 of 3 show the complete ionic and net ionic forms of the following equations. You need to make sure the original equation is balanced before proceeding. He is committed to traditional approaches to knowledge and understanding taught via and in.
This type of reaction is called a precipitation reaction and the solid produced in the reaction is known as the precipitate you can predict whether a precipitate will form using a list of solubility rules such as those found in the table below. Ap topic 12 test b. Adrian has 30 years of high school and early college chemistry teaching experience in both the uk and the usa.
If all species are spectator ions please indicate that no reaction takes place. If the sn2 0 50 m and x3 0 10 m. When two solutions of ionic compounds are mixed a solid may form.
Honors chemistry name period net ionic equation worksheet read this. 2nacl aq pb no 3 2 aq pbcl 2 s 2nano 3 aq. Net ionic equation writing notes.
When two solutions of ionic compounds are mixed a solid may form. Net ionic equation writing test a.

Net Ionic Equations Understanding It Visually Chemistry Lessons Teaching Chemistry Chemistry Classroom

Solubility Rules Doodle Notes Chemistry Worksheets Chemistry Classroom Chemistry Journals

Writing And Balancing Chemical Equations Worksheet In 2020 Chemistry Lessons Teaching Chemistry Chemistry Education

Halogen Reactions Chemical Chemical Chemistry Fluoride

Ionic Equations Net Ionic Equations And Spectator Ions Chemistry Tutorial Chemistry Equations Ionic

Net Ionic Equations Graphic Organizer Concept Map Concept Map Graphic Organizers Chemistry Worksheets
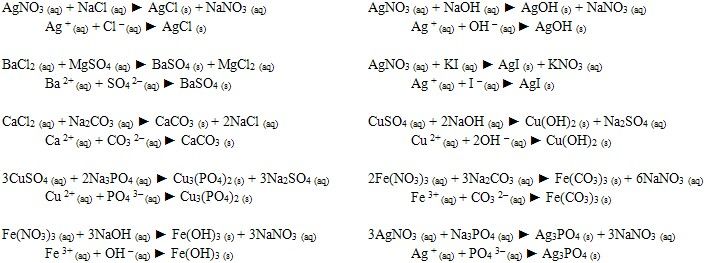
Net Ionic Equations Ap Chem Equations Chemistry Class

Use This 3 Page Worksheet On Balancing Chemical Equations To Show Your Chemistry Students The Differe Chemistry Education Chemistry Lessons Chemistry Classroom

Balancing Equations And Net Ionic Equations For Double Displacement Reactions Science Inquiry Chemistry Balancing Equations

Solubility Rules Doodle Notes High School Chemistry Chemistry Lessons Chemistry Worksheets

Net Ionic Equations Graphic Organizer Concept Map Concept Map Graphic Organizers Equations

Chem121 Precipitation Reaction Spectator Ions And Net Ionic Equations Equations Precipitation Chemistry

Polyatomic Ions Answer Key Pogil Worksheet Template Polyatomic Ion Self Esteem Worksheets

Net Ionic Equations Understanding It Visually Chemistry Worksheets High School Chemistry Equations

Net Ionic Equations Equations Ionic High School Chemistry

Net Ionic Equations Graphic Organizer Concept Map Concept Map Graphic Organizers High School Chemistry

Net Ionic Equations Understanding It Visually High School Chemistry Teaching Chemistry Classroom

How To Write Ionic Equations Tutor Pace Chemistry Notes Science Chemistry Chemistry Class

Complete Ionic Equation Written With All Soluble Strong Electrolytes Shown As Ions Chemistry Education Chemistry Notes Chemistry Lessons

