CuCl2 KOH double replacement 4. Predict the products for and then balance each of the following chemical reactions.

Download Balancing Equations 09 Chemistry Classroom Balancing Equations Chemical Equation
A What is the equilibrium Constant expression for the reaction.

What is a chemical reaction worksheet. Provide a real life example as to how we use temperature to alter. A chemical reaction is described by a chemical equation an expression that gives the identities and quantities of the substances involved in a reaction. PHYSICAL PROPERTY CHEMICAL PROPERTY 1.
4 C 5 H 9 O 29 O 2 20 CO 2 18 H 2 O combustion 4. Further questions just have. Matter will be changed into a new substance after the reaction PHYSICAL AND CHEMICAL PROPERTIES AND CHANGES Name _____.
Chemical and physical changes take place around you all the time. 6CO 2 6H 2 O C 6 H 12 O 6 6O 2 Chemical Reaction for Cellular Respiration. The reactants the starting substances are written on the left and the products the substances found in the chemical reaction are written on the right.
Bond energy is a measure of the strength of a chemical bond. Modification of work by NASA. When you make cereal for breakfast combining the milk and cereal is a physical change.
2Al 3I2 synthesis 3. This worksheet is designed to help you predict products of simple reactions of the four basic reaction types synthesis decomposition single replacement and double replacement and combustion reactions. Answer the following questions using COMPLETE SENTENCES a and b.
A Physical change affects only physical properties ie. A chemical equation shows the starting compoundsthe reactants on the left and the final compoundsthe products on the right separated by an arrow. Exothermic reactions release heat and light into their surroundings.
A chemical change is a permanent change. Indicates how a substance 2. The coefficients next to the symbols of entities indicate the number of moles of a substance produced or used in.
A balanced chemical equation gives the number and type of atoms participating in a reaction the reactants products and direction of the reaction. The answer will appear below. A metal non-metal 2 Na Cl2 2 NaCl sodium chloride Fe S FeS.
Instructions on balancing chemical equations. Determined without destroying matter reacts with something else 2. Observed with senses 1.
Predicting products of chemical reactions – practice problems Directions. Chapter 4 Stoichiometry of Chemical Reactions Figure 41 Many modern rocket fuels are solid mixtures of substances combined in carefully measured amounts and ignited to yield a thrust-generating chemical reaction. Sometimes it can be difficult to tell if a chemical or physical change is taking place.
Chemical reaction compound element endothermic energy exothermic Use the word bank above to fill in the blanks. First FOR ALL EQUILIBRIUM PROBLEMS YOU MUST. Balancing Equations Practice Worksheet.
When atoms rearrange to form new combinations an chemical reaction. Chemical Reactivity Worksheet The Chemical Reactivity Worksheet CRW is a free software program you can use to find out about the chemical reactivity of thousands of common hazardous chemicals compatibility of absorbents and suitability of materials of. It can also be used to demonstrate the chemical changes of color and bubbles.
Fe Au Co Br C O N F. A physical change involves very little to no absorption of energy. PHYSICAL PROPERTY CHEMICAL PROPERTY 1.
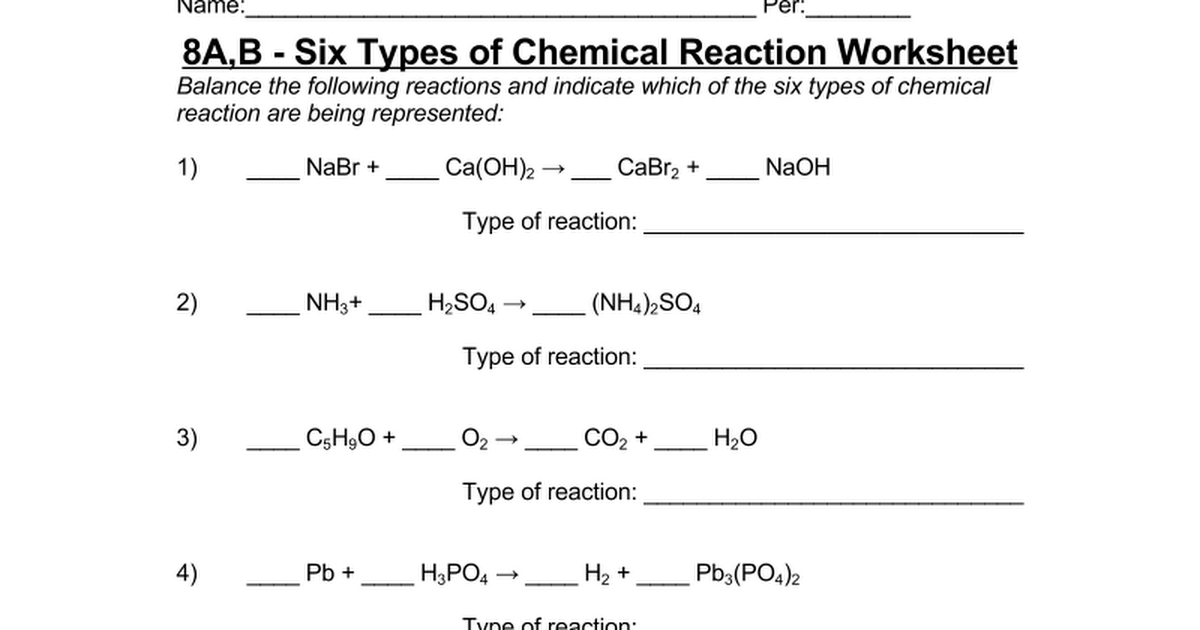
1 Write all equilibrium equations 2 Write all equilibrium concentrations 3 Write all equilibrium expressions SET A. Acid Base Reaction. 2 NaBr CaOH 2 CaBr 2 2 NaOH double displacement 2.
In this article well talk about what a chemical equation is how to balance chemical equations and give you some examples to aid in your balancing. For the first few reactions the type of reaction is listed you should predict the products then balance. C4H10 O2 combustion 7.
During a chemical reaction absorption and evolution of energy. The lab is inquiry and it can be used to introduce the concepts of chemical change. Matter will be changed into a new substance after the reaction PHYSICAL AND CHEMICAL.
Factors Affecting the Rate of Chemical Reactions Worksheet. Enter an equation of a chemical reaction and click Balance. SiI4 Mg single replacement 2.
Always use the upper case for the first character in the element name and the lower case for the second character. The force that holds two atoms together is called an chemical bond. A chemical equation is the symbolic representation of a chemical reaction.
Catalyst and Inhibitors Sometimes a third substance is used in a chemical reaction to speed up or slow down the reaction. CHEMICAL EQUILIBRIUM Name Last Ans. Chemical Reaction for Photosynthesis.
These are really important type of reactions that occur in biological systems. 2 NH 3 H 2 SO 4 NH 4 2 SO 4 synthesis 3. Everything else can be obtained from a grocery store.
Now that you have covered everything that is to be learned about the basics of balancing chemical equations you should get yourself acquainted with certain worthwhile chemical equations. Co – cobalt and CO – carbon monoxide. Mg HCl single replacement 6.
When you eat the cereal a chemical change happens during digestion. Determined without destroying matter reacts with something else 2. The energy change in a chemical reaction is due to the difference in the amounts of stored chemical energy between the products and the reactants.
A balanced chemical equation has the correct number of reactants and products to satisfy the Law of Conservation of Mass. Types of Chemical Reactions Answers Balance each of the following reactions and identify each type of reaction. Photochemical reaction – A photochemical reaction is one involving photons from light.
Observed with senses 1. Such changes generally involve electrons in the highest principle energy level or the. A chemical reaction in which energy is releasedgiven off is _____.
Li 3 N 3. An _____ is a process in which substances are changed into other. Photosynthesis is an example of this kind of chemical reaction.
This is the simple chemical reaction where acid and base are combined together to provide water and salt. 3 Pb 2 H 3 PO 4 3 H 2 Pb 3 PO 4 2 single displacement 5. It is a lab that takes approximatelu 15-20 minutes of class time.
In your own words describe what effect cooling has on the frequency at which particles of reactants can collide. Any reaction in which two or more substances combine to form a single product is a direct union or combination reaction. Chemical Reactions – Ch.
Worksheet – Molar Conversions Worksheet – Molarity Worksheet – Formula Calculations Lab – Molar Conversions Lab – Empirical Formula Lab – Percentage of Water in Popcorn. Balancing an unbalanced equation is mostly a matter of making certain mass and charge are balanced on the reactants and products side of the reaction arrow. This reaction is also called as neutralization reaction and most commonly called as acid-base reaction.
Catalyst Reaction Rate 2. A chemical equation tells you what happens during a chemical reaction. The only limitation would be desk space and graduated cylinders.
This stored chemical energy or heat content of the system is known as its enthalpy. Chemical change both physical and chemical properties of the substance including its composition. An _____ is matter that is composed of 2 or more different kinds of atoms.
Worksheet – Intro to Reactions Worksheet – Balancing Equations Worksheet – Types of Reactions Worksheet – Reaction Energy Rate. The general form of a direct union reaction is A B AB This type of reaction generally takes place between the following types of compounds. Indicates how a substance 2.

2 Pages Types Of Reactions Worksheet Chemistry Worksheets Reaction Types Persuasive Writing Prompts

Balancing Chemical Equations Worksheet Chemistry Worksheets Dimensional Analysis Chemical Equation

Types Of Chemical Reactions Matching Worksheet In Print And Digital Chemistry Worksheets Scientific Method Lesson Plans Scientific Method Lesson

Chemical Reactions Review Color By Answer Worksheet Chemistry Worksheets Biology Worksheet Printable Worksheets

Six Types Of Chemical Reaction Worksheet Types Of Reactions Worksheet In 2021 Chemistry Worksheets Chemical Reactions Reaction Types

Types Of Chemical Reactions Worksheet To You Types Of Chemical Reactions Worksheet Chemistry Worksheets Scientific Method Worksheet Word Problem Worksheets

Classifying And Balancing Chemical Reactions Worksheet Chemistry Worksheets Chemical Reactions Teaching Chemistry

Reaction Types Worksheet Answer Key Pichaglobal Chemistry Worksheets Reaction Types Persuasive Writing Prompts

Balancing Chemical Equations Worksheet Customizable Chemistry Lessons Teaching Chemistry Chemistry Education

Balancing Chemical Equations Worksheets With Answers Chemistry Education Chemistry Lessons Teaching Chemistry

Download Balancing Equations 49 Chemical Equation Equations Balancing Equations
8a B Six Types Of Chemical Reaction Worksheet Word Problem Worksheets Text Evidence Chemistry Worksheets

Use This 2 Page Worksheet Of Chemical Reactions As You Wish As A Homework Assignment A Quiz In 2021 Chemistry Worksheets Chemical Reactions How To Memorize Things

Identify Types Of Chemical Reactions Saferbrowser Yahoo Image Search Results Chemistry Worksheets Chemical Reactions Teaching Chemistry

Customizable And Printable Balancing Chemical Equations Worksheet Teaching Chemistry Chemistry Classroom Chemistry Lessons

Chemical Reactions Types Worksheet Awesome Types Of Chemical Reactions Chemistry Lessons Teaching Chemistry Chemistry Classroom

This Worksheet Can Be Used To Determine How Well Students Understand Balancing Chemical Equations As Well As Identifyi Guided Practice School Related Reactions

Gcse Chemistry Worksheet Balancing Chemical Equations Teaching Resources Chemical Equation Chemistry Worksheets Gcse Chemistry

Do You Find Balancing The Chemical Equation A Daunting Task Download Our Balancing Chemical Equations Workshee Balancing Equations Chemical Equation Equations

