Vsepr valence shell electron pair repulsion is a simple model that employs the concept that electrons being negatively charged are repulsive. Non polar bonds form between elements with similar electronegativities.
H 2 o.

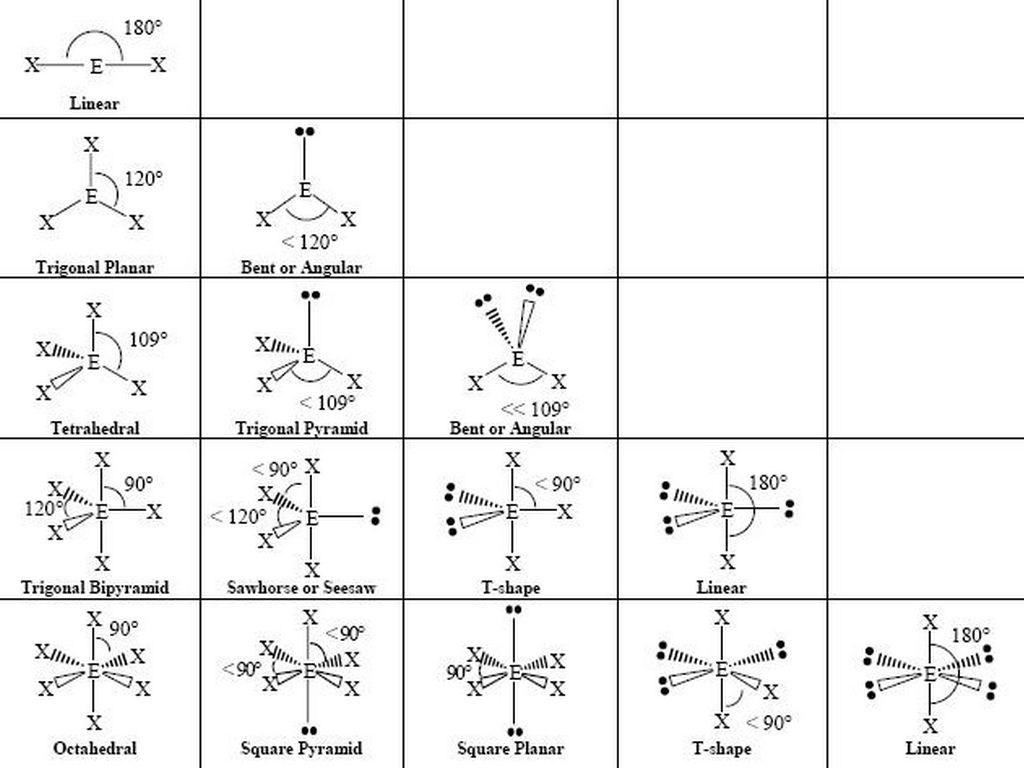
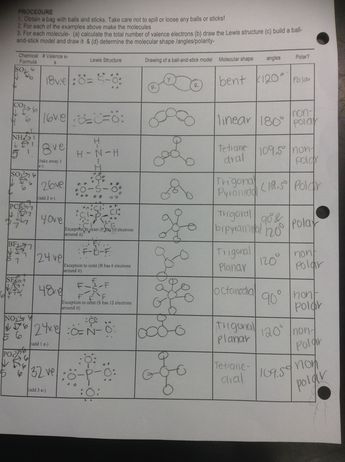
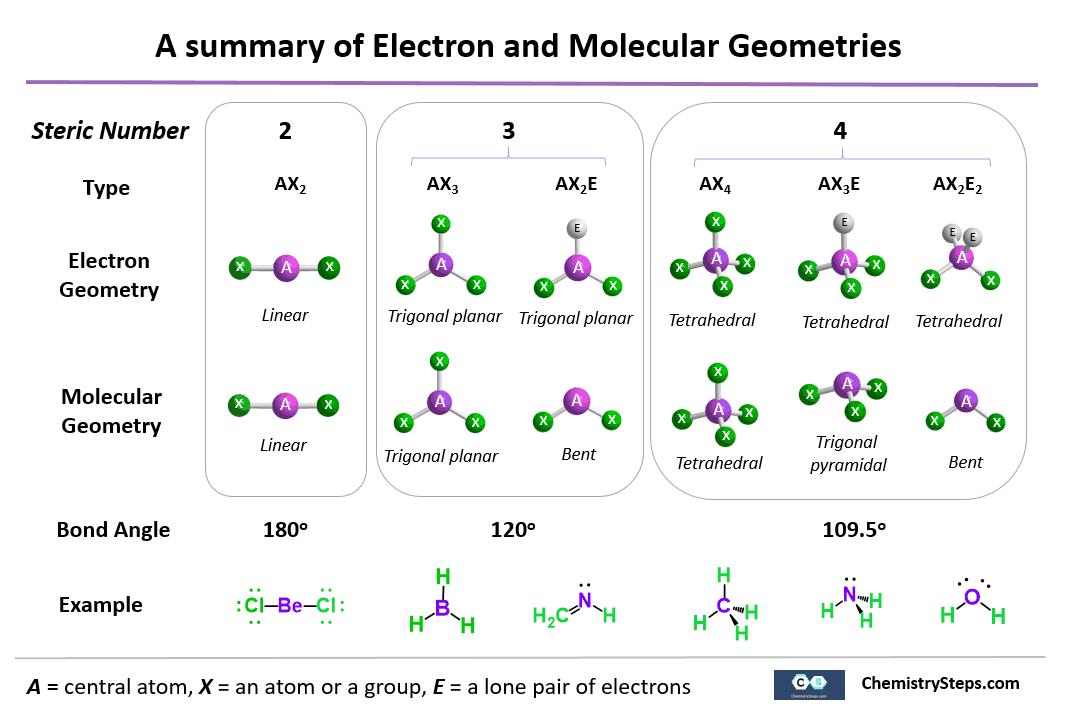
Vsepr and molecular geometry worksheet. An electron group consist of either a single bond a double bond a triple bond or a lone pair of electrons with each counting as one group. Trigonal tetrahedral octahedral bipyramid. Vsepr guidelines for using vsepr theory with lewis structures 1.
Use valence shell electron pair repulsion vsepr model to draw and name molecular shapes bent linear trigonal planar tetrahedral and trigonal pyramidal. Vsepr molecular geometry and polarity resonance and formal charge worksheet i. Vsepr worksheet 1 answers vsepr worksheet 2 answers test review sheet.
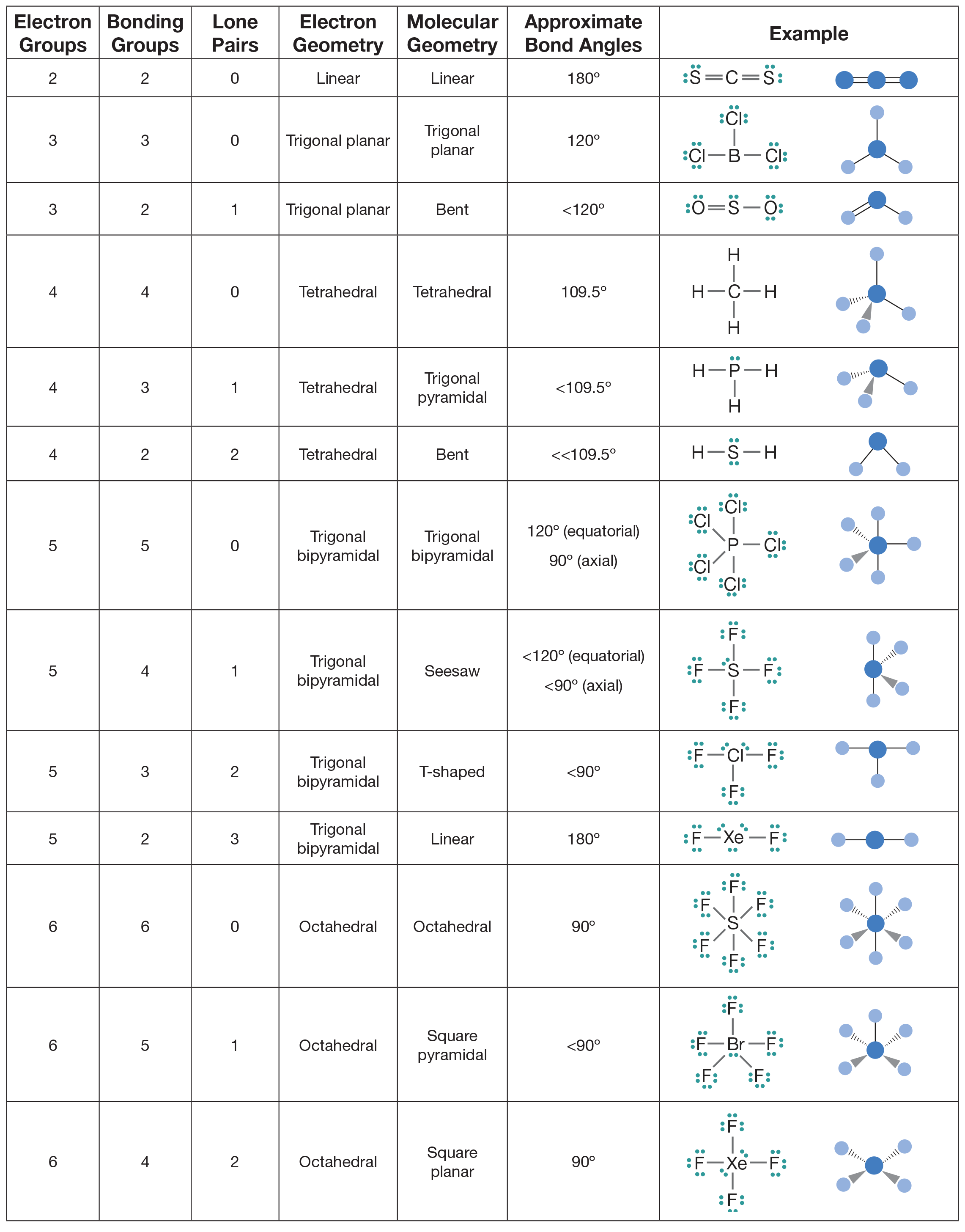
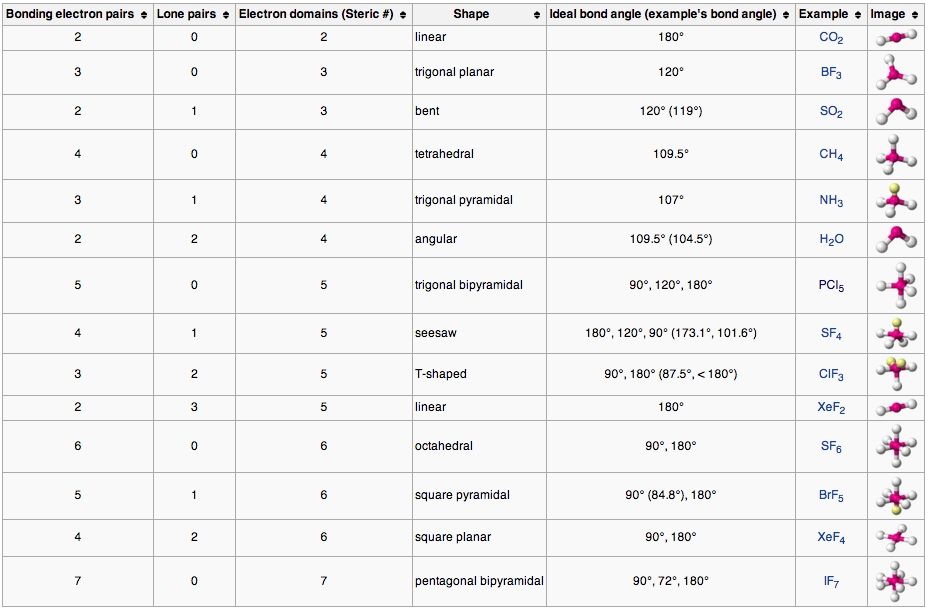
Table of molecular geometries. Polar bonds form between elements with very different electronegativities. Of atoms bonded to central atom of lone pairs of electrons electronic geometry vsepr molecular geometry predicted.
Vsepr worksheets answers self test chapter 10 author s version of a practice test self test chapter 10 answers writing lewis structures for covalent compounds. Worksheet 13 molecular shapes the shapes of molecules can be predicted from their lewis structures by using the vsepr valence shell electron pair repulsion model which states that electron pairs around a central atoms will assume a geometry that keeps them as far apart from each other as possible. Therefore regions of electron densities will.
The valence shell electron pair repulsion vsepr theory or vesper for short is how the geometry of a molecule is determined around a central atom. Molecular geometry hbr linear nh 3. So 4 2 tetrahedral.
Electronic geometry and vsepr molecular geometry. Ap chemistry ap chemistry quizzes. Test is friday february 06 2015.
Molecular geometry vsepr at this point we are ready to explore the three dimensional structure of simple molecular covalent compounds and polyatomic ions. Angles example molecule hybridization 2 0 linear linear 180 becl2 sp. We will use a model called the valence shell electron pair repulsion vsepr model that is based on the repulsive behavior of electron pairs.
This is illustrated by the drawings below. This quiz involves chemical bonds and the structure and geometry of molecules including lewis structures and vsepr theory. No 3 1 triangular.
The molecular geometry main shapes are tetrahedral trigonal planar trigonal pyramidal bent and linear and are named by measuring the bond angles between the central atom and another atom. To determine a geometry first construct a lewis structure then consult the table below. Po 4 3 tetrahedral.
Vsepr Theory Ppt Vsepr Theory High School Chemistry Molecular Geometry
Vsepr Theory Molecular And Electron Geometry Of Organic Molecules Chemistry Steps In 2020 Vsepr Theory Molecular Geometry Organic Molecules

Vsepr Theory And Molecular Geometry Vsepr Theory Molecular Geometry Chemistry Worksheets

Mol Geom Gif Vsepr Theory Molecular Geometry High School Chemistry

Structural Biochemistry Molecular Geometry Wikibooks Open Books Molecular Geometry Vsepr Theory Chemistry Textbook

Molecular Geometry And Vsepr Molecular Geometry Molecular Chemistry

A Nice Infographic Table About The Vsepr Guidelines And Geometry Around Atoms Teaching Chemistry Chemistry Classroom Molecular Geometry

Molecular Geometry Vsepr Molecular Geometry Chemistry Worksheets Chemistry

Molecular Structure By Vsepr Molecular Geometry Molecular Molecular Shapes
Electron And Molecular Geometries Molecular Geometry Teaching Chemistry Chemistry Lessons

Difference Between Electron Geometry And Molecular Geometry Definition Identification Examples Molecular Geometry Vsepr Theory Chemistry Textbook

How To Predit Polarity Of Molecules Biochemhelp Molecular Geometry Teaching Chemistry Chemistry Help

Molecular Structure Chart Geometries Anta Co Geometry Pdf Molecular Geometry Molecular Molecular Structure

Vsepr Theory Molecular Geometry Organic Chemistry Geometry Worksheets

4 Molecular Geometry Worksheet In 2020 Molecular Geometry Geometry Worksheets Molecular
Pin By Mara Tu On Chemistry Molecular Geometry Chemistry Chemistry Classroom

Vsepr Theory And Molecular Geometry Worksheet And Cheat Sheet By Omg Science In 2020 Molecular Geometry Vsepr Theory Geometry Worksheets

Pin By Jessica L Santos On Chemistry Vsepr Theory Chemistry Classroom Organic Chemistry Study

How To Predict Polarity Of Molecules Based On Their Shape Biochemhelp Molecular Geometry Chemistry Classroom Chemistry Lessons