In this video we cover. Balancing redox reactions in base.

Tang 02 Balancing Redox Reactions 2 Lezioni Di Scienze Chimica Scienza
P cu 2 æ cu h2po4 ph3 i2 æ h3po2 i no2 æ no3 no.

Redox reactions worksheet gcse. In the reaction cl2 2br aq 2cl aq br2 which half reaction correctly represents oxidation. This website and its content is subject to our terms and conditions. Oxidizing and reducing agents.
Oxidation and reduction in terms of oxygen and electrons redox reactions displacement reactions ionic equations half equati. Mn 2 bio3 æ mno4 bi 3 mno4 s2o3 2 æ s4o6 2 mn 2 clo3 cl æ cl2 clo2. Balancing redox reactions in acid.
Balancing redox reactions worksheet 1 balance each redox reaction in. Clo2 æ clo2. A quick sheet to assess student understanding of the processes.
Identify the pair of elements undergoing oxidation and reduction by checking oxidation states. Write two ionic half equations one for the oxidation one for the reduction balance elements other than o and h then balance charges by adding electrons. Combustion that occurs in such a way that fuel is not completely oxidized burned up.
H 2o 2 cr 2o 7 2 o 2 cr 3 9. This is excellent a really useful worksheet. The transfer of protons d.
The steps for balancing redox reactions in basic solution are. Our mission is to provide a free world class education to anyone anywhere. Donate or volunteer today.
Can they identify the various key points given an equation. Mno 2 mn 2o 3 balance each redox reaction in acid solution using the half reaction method. Teo 3 2 n 2o 4 te no 3 10.
Divide the reaction into half reactions clo 3 aq cl aq i aq i 2 s step 2. A change in phase c. No no 3 6.
Gcse chemistry redox reactions equation practice. Cr 2o 7 2 cr3 5. Mno4 c2o4 2 æ mno2 co2.
A change in oxidation number b. Additional notes redox reaction both oxidation and reduction going on side by side combustion oxidation that occurs so rapidly that noticeable heat and light are produced. Oxidizing and reducing agents.
Khan academy is a 501 c 3 nonprofit organization. Worksheet 5 balancing redox reactions in acid and basic solution balance each half reaction in basic solution. A redox reaction always involves a.
Balance atoms and charges in each half reaction atoms other than o and h clo 3 aq cl aq cl is balanced 2i aq i 2 s i now balanced balance o atoms by adding h 2 o molecules clo 3 aq cl aq 3h 2 o l add 3h 2 o 2i aq i 2 s no change. So 4 2 so 2 7. The formation of ions 17.

Picture Redox Reactions Free Math Worksheets Chemistry Notes

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Lessons Chemistry Education

Describing Redox Reactions Handout And Worksheet Redox Reactions Electrochemistry Science Chemistry
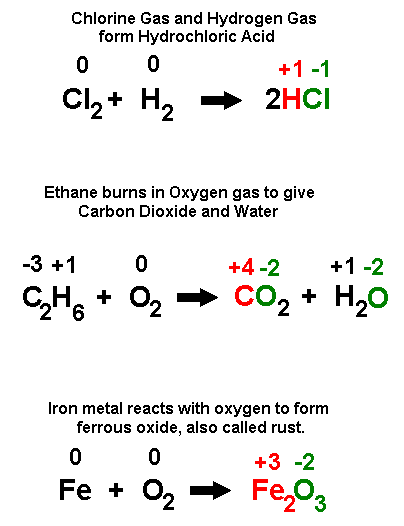
Oxidation Reduction Reactions Oxidation Involves The Loss Of Electrons While Reduction Involves The Teaching Chemistry Chemistry Classroom Chemistry Education

Students Will Be Practicing Writing Half Reactions For A Redox Reaction By Completing This Practice Works Practices Worksheets Writing Practice Redox Reactions

Ncert Solutions For Class 11 Chemistry Chapter 8 Redox Reactions 026 Redox Reactions 11th Chemistry Chemistry

Oxidation Reduction Guide Answers Chemistry Worksheets Chemistry Lessons Oxidation

Balancing Chemical Equations Worksheets With Answers In 2020 Balancing Equations Chemical Equation Equations

Tang 02 Balancing Redox Reactions 2 Redox Reactions Organic Reactions Reactions

Worksheet For Balancing Chemical Equations Chemical Equation Balancing Equations Chemistry Worksheets

Examples Of Reduction Reactions Chemistry High School Chemistry Science Lessons

Describing Redox Reactions Handout And Worksheet Redox Reactions Electrochemistry Science Chemistry

Learn How To Recognize Redox Reactions Oxidation Species Reduction Species And Half Reactions Mcat Chemist Redox Reactions Ap Chemistry Chemistry Notes

Redox Reactions Exercise With Solutions Redox Reactions Chemical Equation Reactions

Organic Chemistry Infographic Redox Reactions In Bio Organic Chemistry Organic Chemistry Chemistry Teaching Chemistry

Oxidation Reduction Worksheet Answers 25 Redox Review Worksheet Answers In 2020 Scientific Notation Word Problems Word Problem Worksheets Pre Algebra Worksheets

Pin By Luci D Amico On Balancing Of Chemical Reaction Redox Reactions Chemistry Worksheets Chemistry Lessons

Picture Redox Reactions Chemistry Class Equations

Redox Reaction Redox Reactions Teaching Chemistry Chemistry Notes

