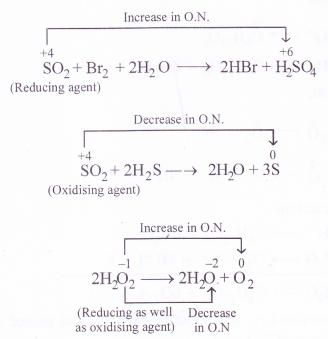
Fe 2 o 3 3 co 2 fe 3 co 2 in acidic solution c. Write the redox reaction and predict the spontaneity for the following.
Chemistry Redox Reaction Notes Interactive Redox Reactions Organic Chemistry Study Chemistry Notes
Our mission is to provide a free world class education to anyone anywhere.

Recognizing redox reactions worksheet. In which substance is the oxidation number of nitrogen zero. Write balanced equations for the following redox reactions. Divide the skeleton reaction into two half reactions each of which contains the oxidized and reduced forms of one of the species 2.
Oxidizing and reducing agents. What is the oxidation number of carbon in nahco3. Reactions in which only one chemical is both oxidized and reduced at the same time are called disproportionation.
In the reaction al0 cr3 al3 cr0 the reducing agent is a. Because there is a change in oxidation number we can confidently say that the above equation represents a redox reaction. Once you recognize that you will notice that cu is both oxidized to cu 2 and reduced to cu.
Cr sn 4 cr 3 sn 2 b. Some species are on both sides of the redox table and may act as either oa or ra. In each case assume that the reaction takes place in an acidic solution also state whether the reaction is oxidation or reduction.
3 hg 2 2 fe s 3 hg 2 2 fe 3 c. Oxidizing and reducing agents. H 2 co 3 b.
Atoms other than o and h then o then h. Fe 3 o 4 hint. Donate or volunteer today.
2 nabr cl 2 2 nacl br 2 b. Balancing redox reactions in base. Consider the following reactions.
A concentrated nitrous acid is poured on to a strip of zinc. 5 co i 2 o 5 5 co 2 i 2 in basic solution. Balancing redox reactions worksheet 1 balance each redox reaction in.
Redox practice worksheet name. Khan academy is a 501 c 3 nonprofit organization. Determine the oxidation number of the elements in each of the following compounds.
Predicting redox reactions using the half reaction table 1. Ws 4 balancing redox reactions. Cr oh 3 br 2 cro 4 2 br in basic solution 10 oh 2 cr oh 3 3 br 2 2 cro 4 2 8 h 2 o.
Which one resembles a redox reaction. Identify the species being oxidized and reduced in each of the following reactions. Mn 2 bio3 æ mno4 bi 3 mno4 s2o3 2 æ s4o6 2 mn 2.
There should be an oxidizing species and a reducing species. Write balanced equations for the following reactions. Zn s cuso 4 aq znso 4 aq cu s hcl aq naoh aq nacl aq h 2 o l in a redox reaction oxidation numbers change in reactants and products.
Balancing redox equations method 2. In the reaction 2k cl2 2kcl the species. Balance the atoms and charges in each half reaction atoms are balanced in order.
O h 2o l o sn 2 aq o cr2 aq o fe2 aq worksheet. Balancing redox reactions in acid. Balance each of the following half cell reactions.

Picture Redox Reactions Free Math Worksheets Chemistry Notes

Chemistry Multiple Multiple Choice Types Of Chemical Reactions Chemistry Chemical Reactions Multiple Choice

Redox Reactions Cbse Notes For Class 11 Chemistry Learn Cbse Cbsenotesclass11chemistry Redoxreactions 11th Chemistry Redox Reactions Chemistry Notes

Chemical Reactions Types Worksheet Unique 16 Best Of Types Chemical Reactions Worksheets Chessmu In 2020 Reaction Types Persuasive Writing Prompts Chemical Reactions

Triominos Puzzle Gas Laws Review Chemistry Chemistry Electrochemistry Fun Science

Illustrated Glossary Of Organic Chemistry Redox Reaction Chemistry Study Guide Chemistry Classroom Redox Reactions

Pin By Kristen Hicks On Teaching Ideas Teaching Chemistry Chemistry Teaching

Shops 1 20 Of 5004 Life Science Middle School 7th Grade Science Science Worksheets

Worksheet For Balancing Chemical Equations Chemical Equation Balancing Equations Chemistry Worksheets

Learn How To Recognize Redox Reactions Oxidation Species Reduction Species And Half Reactions Mcat Chemist Redox Reactions Ap Chemistry Chemistry Notes

Balancing Chemical Equations Questions A Process Chemical Equation Balancing Equations Chemistry Chemical Equations Chemistry

What Is Electrolysis And Electrolyte With Examples In 2020 Redox Reactions Chemistry Chemistry Free

Arrhenius Bronsted Lowry And Lewis Acids And Bases In Organic Chemistry Understand The Simil Organic Chemistry Study Chemistry Education Teaching Chemistry

Grades 10 12 Redox Balancing Redox Reactions Practice Worksheet In This Hands On Worksheet Students Will Redox Reactions Reactions Practices Worksheets

Chemical Reactions Types Worksheet Awesome Types Of Chemical Reactions In 2020 Chemistry Lessons Teaching Chemistry Science Chemistry

Oxidation Reduction Chemistry Education Chemistry Lessons Teaching Chemistry

Redox Identifying The Oxidizing And Reducing Agents Practice Worksheet Practices Worksheets Reducing Agent Worksheets