Mg mg2 2e d. The reaction that takes place in a chemical cell is best classi ed as a.
Pin By Saitech Informatics On Balancing Of Chemical Reaction Equations Distance Time Graphs Worksheets Redox Reactions
2cs br2 2csbr 4.

Recognizing redox reactions worksheet answers. In the reaction mg cl2 mgcl2 the correct half reaction for the oxidation that occurs is a. 3mg n2 mg3n2 5. If the redox reaction was carried out in basic solution i e.
Reduction and oxidation by gain and loss in oxygen reduction and oxidation by gain and loss of electrons reduction and oxidation by gain and loss of electron numbers redox of complex ions. 5 co i 2 o 5 5 co 2 i 2 in basic solution. Atoms other than o and h then o then h.
Determine what is oxidized and what is reduced in each reaction. Cl2 2e 2cl c. Redox reactions are a chemical reaction in which electrons are exchanged through oxidation and reduction.
Determination of activity of some metals by reaction with hydrogen ion doc 28 kb redox reactions docx 18 kb half reactions and balancing warm up doc 51 kb balancing half reactions worksheet doc 31 kb balancing redox equations docx 16 kb electrochemical cell diagrams i ii doc 230 kb electrochemical cell diagrams iii iv doc 230 kb. Write balanced equations for the following reactions. The steps for balancing redox reactions in basic solution are.
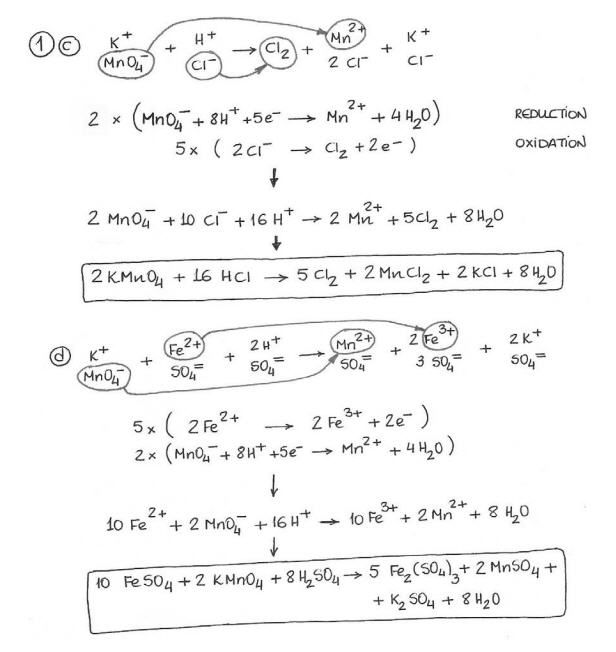
Which equation represents the half reaction that takes place at. Ws 4 balancing redox reactions. Divide the skeleton reaction into two half reactions each of which contains the oxidized and reduced forms of one of the species 2.
In each case assume that the reaction takes place in an acidic solution also state whether the reaction is oxidation or reduction. Write balanced equations for the following redox reactions. Fe 2 o 3 3 co 2 fe 3 co 2 in acidic solution c.
Balancing redox reactions in basic solution. Oxidising and reducing agents. 2 worksheets consisting over 70 questions and answers of topics related to.
2ca o2 2cao 9. A concentrated nitrous acid is poured on to a strip of zinc. Predicting redox reactions using the half reaction table 1.
Write the redox reaction and predict the spontaneity for the following. 4fe 3o2 2fe2o3 6. Cr oh 3 br 2 cro 4 2 br in basic solution 10 oh 2 cr oh 3 3 br 2 2 cro 4 2 8 h 2 o.
Identify the pair of elements undergoing oxidation and reduction by checking oxidation states. Balancing redox equations method 2. About this quiz worksheet.
Suited for student in y10 and y11. Balance each of the following half cell reactions. Balance the atoms and charges in each half reaction atoms are balanced in order.
Alkaline conditions then we have to put in an extra step to balance the equation. 2sr o2 2sro 2. This quiz and worksheet have been assembled to test your.
Cl2 2nabr 2nacl br2 7. Identify the oxidizing agent and the reducing agent also. O h 2o l o sn 2 aq o cr2 aq o fe2 aq worksheet.
Some species are on both sides of the redox table and may act as either oa or ra. 2li s li2s 3. 2 nabr cl 2 2 nacl br 2 b.
Si 2f2 sif4 8.

Describing Redox Reactions Handout And Worksheet Redox Reactions Electrochemistry Science Chemistry

Quiz Worksheet How To Balance Redox Reactions And Identify Oxidizing And Reducing Agents Study Com

Chemical Reactions Types Worksheet Awesome Types Of Chemical Reactions In 2020 Chemistry Lessons Teaching Chemistry Science Chemistry

4 6 Precipitation Reactions Writing Equations Compound Words Precipitation

Oxidation Reduction Reaction Lesson Plans Worksheets

Worksheet A Organic Redox Reactions Certain Organic Compounds

Redox Rxn Wksht 11 1 Worksheet 1 1 Name Redox Reactions Period 1 What Is A Redox Reaction 2 Can Oxidation Occur Without Reduction Explain 3 Why Would Course Hero

Worksheet 7 Oxidation Reduction Reactions Oxidation Number

Chemical Reactions Types Worksheet Unique 16 Best Of Types Chemical Reactions Worksheets Chessmu In 2020 Reaction Types Persuasive Writing Prompts Chemical Reactions

Oxidation Numbers And Redox Reactions Yourhomework Com

As Chemistry Redox Reactions And Group 2 Elements Chemistry Classroom Teaching Chemistry Science Chemistry

Tang 02 Balancing Redox Reactions Redox Reactions Teaching Chemistry Teaching Science

Picture Redox Reactions Free Math Worksheets Chemistry Notes

Oxidation Reduction Reaction Lesson Plans Worksheets Lesson Planet

Redox Reactions Concept Map Mtg Chemistry Today Magazine Jeemain Jeeadvanced Class11 Cl Chemistry Classroom Chemistry Lessons Organic Chemistry





