Oxidation reduction packet. This method relates the number of electrons transferred to change in oxidation number.

Tang 02 Balancing Redox Reactions 2 Lezioni Di Scienze Chimica Scienza
Cr 2o 7 2 c2h 4o c 2h 4o 2 cr 3 4.

Oxidation reduction worksheet pdf. Identify the changes in oxidation states and write the oxidation half reaction and the reduction half reaction and for each half reaction. Mno 2 mn 2o 3 19. Give the oxidation number of each kind of atom or ion.
Hclo 4 hcl h 2o 21. The oxidation state of iron decreases from 3 in fe 2 o 3 to zero in the uncombined element fe. Use the changes in oxidation numbers to determine which elements are oxidized and which.
Zn no 3 zn2 nh4 3. Page 2 redox practice worksheet. Gain electrons only b.
Oxidation cannot occur without reduction. Ptcl 6 2 c. The oxidation state of carbon increases from 2 in co to 4.
Remember that if the oxidation increases it means oxidation and when it decreases it mean reduction. Both gain and. Microsoft word 14 04 oxidation numbers worksheet doc author.
Ag no 3 ag no 2. Oxidation numbers are either real charges or formal charges which help chemists keep track of electron transfer. Give oxidation numbers for the underlined atoms in these molecules and ions.
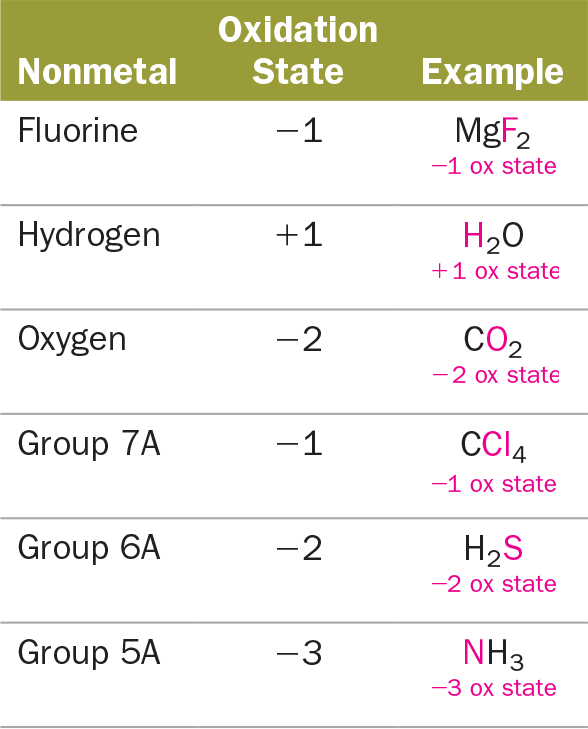
Oxidation reduction redox reactions are reactions in which oxidation numbers change. In practice oxidation numbers are best viewed as a bookkeeping device. O 2 o2 22.
Balance the atom undergoing redox changes if necessary. H 2so 4 22. State of the change that represents oxidation reduction or neither.
Calculate the oxidation number of chromium in each of the following. H 3po 2 cr 2o 7 2 h3po 4 cr 3 basic solution. In the reaction agno3 aq nacl aq nano3 aq agcl s the reactants a.
Al 2o 3 f. Ptcl 4 2 n. Both oxidation and reduction d.
Mno 4 m. Lose electrons only c. Oxidation reduction handout the original concept of oxidation applied to reactions where there was a union with oxygen.
The oxidation state of oxygen is 2 in all compounds therefore oxygen is neither oxidized or reduced. Neither oxidation nor reduction 23. P 2o 5 p 4h 10 determine the oxidation number 23.
Oxidation reduction balancing additional practice problems acidic solution 1. Sbf 6 i. Likewise then reduction applied to reactions where there was a removal of oxygen.
Nh 3 no 2 20. In the reaction 3cl2 6naoh 5nacl naclo3 3h2o. Therefore iron gains electrons and fe 2 o 3 is the oxidizing agent.
The oxygen was either furnished by elemental oxygen or by compounds containing oxygen. H 3aso 3 h. It consists of the following steps.
4 9 Oxidation Reduction Reactions Chemistry Oxidation Reactions

Electrochemistry Notes And Galvanic Cell Notes Galvanic Cell Chemistry Notes Redox Reactions

Oxidation Reduction Reactions 10th Higher Ed Worksheet Scientific Notation Word Problems Factoring Quadratics Word Problem Worksheets

Ncert Solutions For Class 11 Chemistry Chapter 8 Redox Reactions 012 11th Chemistry Chemistry Redox Reactions

How To Assign Oxidation Numbers In 2020 Reducing Agent Oxidation Redox Reactions

Ncert Solutions For Class 11 Chemistry Chapter 8 Redox Reactions 025 Redox Reactions 11th Chemistry Chemistry

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Lessons Chemistry Education
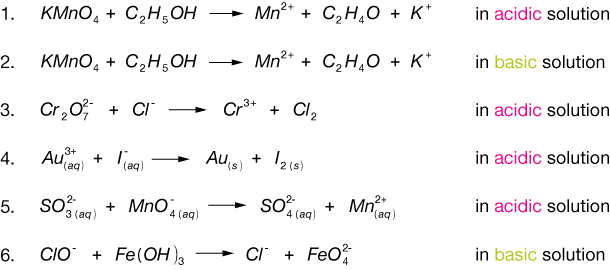
Redox Balancing Redox Reactions Chemistry Lagrange College

This Puzzle Is A Great Review Of Oxidation X2f Reduction Voltaic Cells And General Terms Related To Electrochemistry Teaching Chemistry Chemistry Education

Redox Reactions Redox Reactions Chemistry Education Chemistry Worksheets

Redox Identifying The Oxidizing And Reducing Agents Practice Worksheet Practices Worksheets Reducing Agent Worksheets

Learn How To Recognize Redox Reactions Oxidation Species Reduction Species And Half Reactions Mcat Chemist Redox Reactions Ap Chemistry Chemistry Notes

Chemistry Notes Chemistry Pdf Electrochemistry And Galvanic Cells In 2020 Electrochemistry Chemistry Notes Chemistry

Properties Of Addition Worksheets Pdf Property Addition Worksheets Worksheet Addit In 2020 Addition Worksheets Properties Of Addition Multi Step Equations Worksheets

Oxidation Reduction Reactions Oxidation Involves The Loss Of Electrons While Reduction Involves The Teaching Chemistry Chemistry Classroom Chemistry Education

Oxidation Reduction Guide Answers Chemistry Worksheets Chemistry Lessons Oxidation

Pin By Saitech Informatics On Balancing Of Chemical Reaction Equations Distance Time Graphs Worksheets Redox Reactions

Oxidation Reduction Redox Reactions Balancing Redox Reactions Chemistry Net Redox Reactions Teaching Chemistry Chemistry Lessons