Write the symbols for each element. Ionic bonding in covalent bonding the valence electrons are shared as pairs between the bonded atoms.
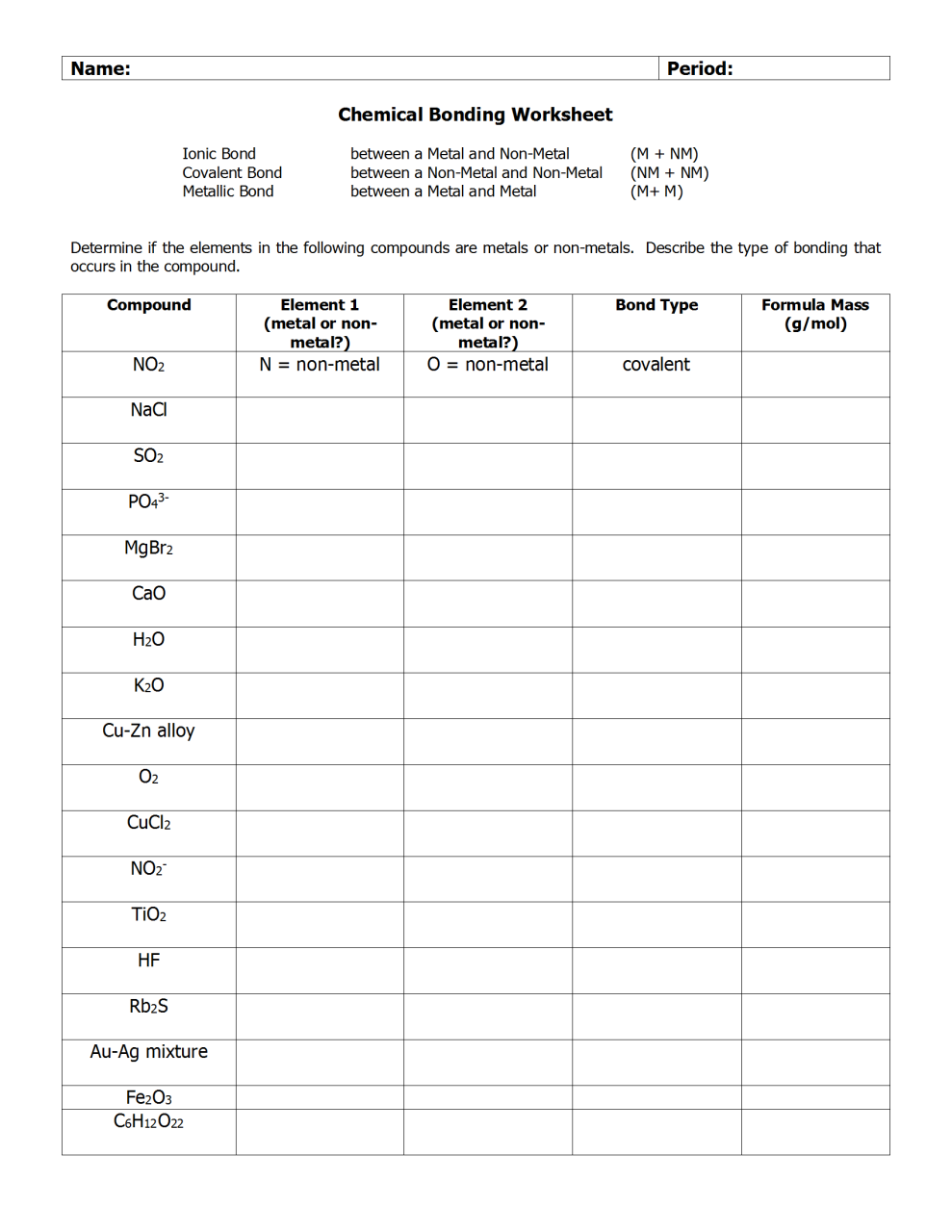
Bonding Worksheet Doc Covalent Bonding Worksheet Covalent Bonding Ionic Bonding
Previous to dealing with bonding basics ionic bonds worksheet answers be sure to understand that knowledge will be your crucial for a much better the next day and also finding out doesn t only quit after a institution bell rings that will being claimed most of us provide you with a selection of uncomplicated however informative articles or blog posts as well as layouts built ideal for.

Ionic bonding worksheet pdf. Show how covalent bonding occurs in each of the following pairs of atoms. Fill in the missing information on the chart. Covalent bonding name covalent bonding occurs when two or more nonmetals share electrons.
On this dedicated page you will be able to revise ionic bonds through our ionic bonding revision worksheet and resource links. Ionic bond between a metal and non metal m nm covalent bond between a non metal and non metal nm nm determine if the elements in the following compounds are metals or non metals. For example when sodium metal and chlorine gas are brought into contact they react violently and we obtain sodium chloride.
Assume in the first instance that compounds between reactive metals and reactive non metals will be ionic. Ions are formed where electrons are transferred from the valence shell of one atom usually a metal to the valence shell of another atom non metal so that both end up with noble gas configurations. Solutions for the naming ionic compounds practice worksheet 1 ammonium chloride 2 iron iii nitrate 3 titanium iii bromide 4 copper i phosphide 5 tin iv selenide 6 gallium arsenide 7 lead iv sulfate 8 beryllium bicarbonate 9 manganese iii sulfite 10 aluminum cyanide 11 cr po 4 2 12 v co.
In ionic bonding the valence electrons are com pletely transferred from one atom to the other atom. Describe the type of bonding that occurs in the compound. Whether you are sitting aqa gcse chemistry or edexcel combined science you need to know about ionic bonds.
Ionic bonds occur between metals and nonmetals when there is a large difference in electronegativity. This reaction is shown below. Element of protons of electrons of valence electrons sodium chlorine beryllium fluorine lithium oxygen phosphorus 2.
7 2 ionic bonding the chemical bond formed by transfer of electron from a metal to a non metal is known as ionic or electrovalent bond. 2na s cl 2 g 2nacl s. Ionic bonds practice 1.
Ionic bonding is a staple part pf every gcse chemistry syllabus. Attempting to attain a stable octet of electrons at least part of the time. For each of the following ionic bonds.
Note that hydrogen is content with 2 not 8.

This Activity Is A Way To Practice Classifying Chemical Formulas As Either Ionic Or Covalent Bonds Covalent Bonding Teaching Chemistry Ionic And Covalent Bonds

Ionic And Covalent Bonding Practice Worksheet Answers Worksheets

Naming Covalent Compounds Worksheet Naming Binary Covalent Compounds Naming Compounds Worksheet Grief Worksheets Covalent Bonding Worksheet

Naming Ionic And Covalent Compounds Worksheet Google Search Ionic Compound Practices Worksheets Naming Chemical Compounds Worksheet

Ionic Vs Covalent Coloring Activity Chemistry Science Pdf Printable From Laurelsusanstudio On Teacher Chemistry Lessons Teaching Chemistry Chemistry Classroom

Ionic Vs Covalent Coloring Activity Chemistry Science Pdf Printable From Laurelsusanstudio On Teache Chemistry Activities Teaching Chemistry Chemistry Lessons

Chemical Bonding Cheat Sheet How To Name Ionic And Covale Ionic And Covalent Bonds Covalent Bonding Chemistry Worksheets

This Could Be Used As An Assessment Or Practice Worksheet Over Chemical Bonding Covalent Bonding Worksheet Chemistry Worksheets Covalent Bonding

Ionic Compounds Names And Formulas Worksheet November 17 2017 Covalent Bonding Worksheet Practices Worksheets Covalent Bonding

Ionic Bonding Note November 14 2017 Covalent Bonding Worksheet Ionic Bonding Covalent Bonding

Types Of Chemical Bonds Cute Google Search
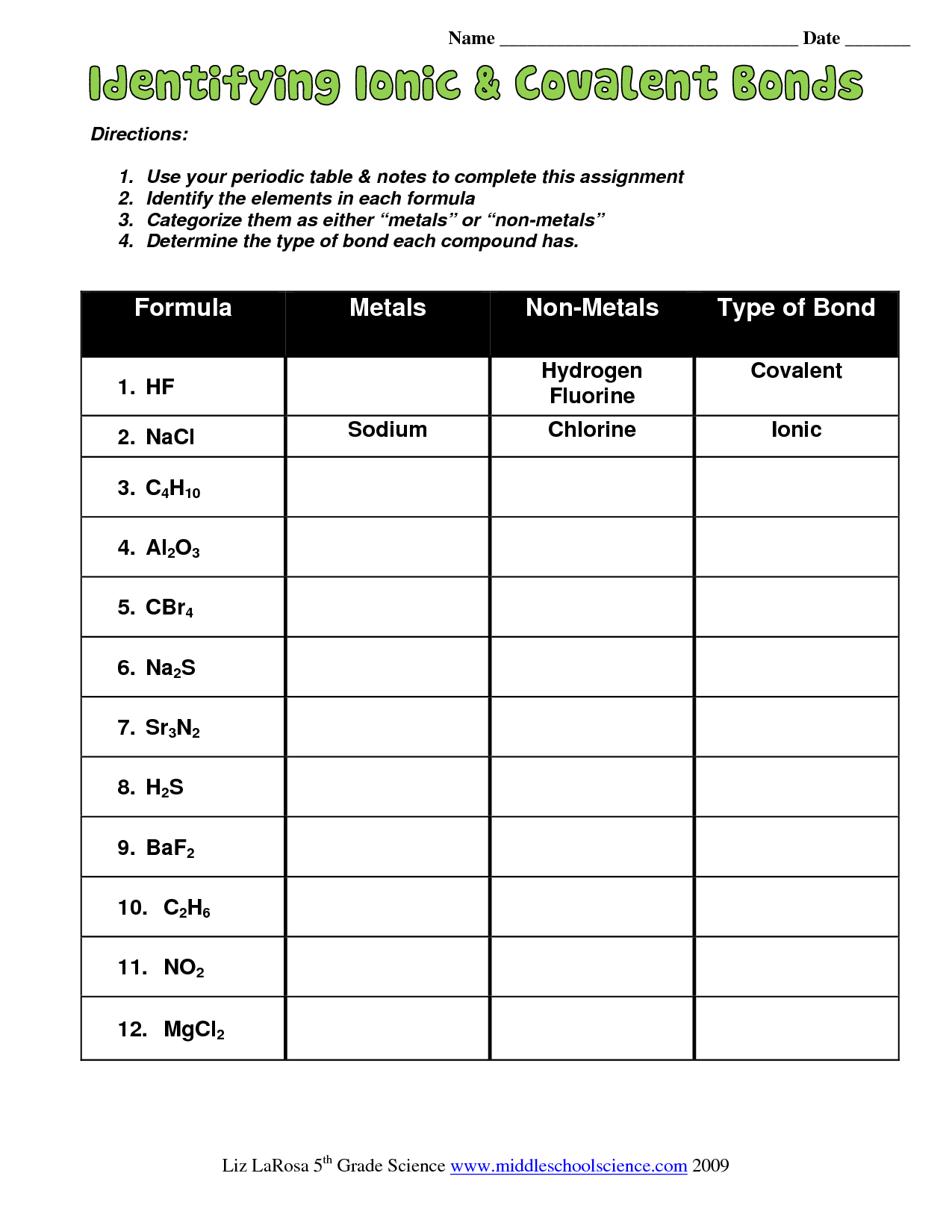
Igcse Identifying Ionic Covalent Bonds Covalent Bonding Ionic And Covalent Bonds Chemistry Worksheets

Ionic Bonds Student Exploration Gizmo Worksheet Ionic Bonding Covalent Bonding Worksheet Covalent Bonding

Ionic Bonding Note November 14 2017 Covalent Bonding Worksheet Ionic Bonding Covalent Bonding
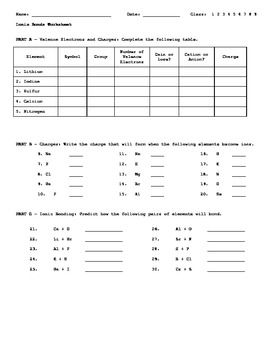
Ionic Bonds Worksheet Chemistry Worksheets Ionic Bonding Ionic And Covalent Bonds

Covalent Bonding Worksheet Answers Teaching Chemistry Chemistry Lessons Chemistry Classroom

Covalent Bonding Worksheet Covalent Bonding Worksheet Covalent Bonding Ionic Bonding

Practice With Covalent Bonding Covalent Bonding Covalent Bonding Worksheet Teaching Chemistry


