The combined gas law states that a gas pressure volume temperature constant. K and then i raise the pressure to 14 0 atm and increase the temperature to 300.
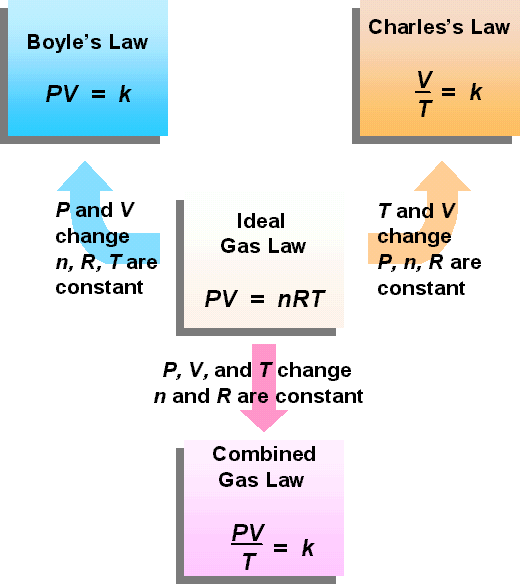
Reich Chemistry The Combined Gas Law Bertino Chemistry Lessons Teaching Chemistry Chemistry Education
Gas laws worksheet 1 bo le s charles ga lussac s and combined gas law solve all problems you must show your work including units.

Combined gas law worksheet answer key. What will its volume be at 20 0 c and 780 0 mm of mercury pressure. As always include enough work and show the units to ensure full credit. 262 5 mm hg 6.
A helium filled balloon at sea level has a volume of 2 1 liters at 0 998 atm and 36 c. Gas laws worksheet atm 760 0 mm hg 101 3 kpa 760 0 torr boyle s law problems. The correct answer is given in parentheses at the end of the problem.
Combined gas law worksheet 1. Combined gas law problems 1 a sample of sulfur dioxide occupies a volume of 652 ml at 40 c and 720 mm hg. Combined gas law and avogadro s principle 18.
Use the combined gas law to solve the following problems. What is the new volume. 2 a sample of argon has a volume of 5 0 dm3 and the pressure is 0 92 atm.
Solve the following problems. It was developed in 1990 to provide people with the answer key to questions in lawlor s gas law program. Combined gas law worksheet answer key is a computer program developed by researcher robert lawlor.
What volume will the sulfur dioxide occupy at stp. If 22 5 l of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. Combined gas law the combined gas law combines charles law boyle s law and gay lussac s law.
The combined law for gases. Calculate the ratio of effusion rates for nitrogen n 2 and neon ne. K what is the new volume of the gas.
The combined gas law key. A as ple contained in a cylinder equipped with a moveable piston occupie 00 0 at a pressure. 1 if i initially have a gas at a pressure of 10 0 atm a volume of 24 0 liters and a temperature of 200.
1 atm 760 0 mm hg 101 3 kpa k 273 oc a gas balloon has a volume of 106 0 liters when the temperature is 45 0 c and the pressure is 740 0 mm of mercury. Gas laws worksheet 2 08 modified 3 17 answer key graham s law 1. If the final temperature is 30 c the final volume is 5 7 l and the final.
A gas with a volume of 4 0l at a pressure of 205kpa is allowed to expand to a volume of 12 0l. The pressure of a gas changes from 120 kpa to 50 kpa. Combined gas law problems.
Boyle s law problems charles law problems guy lussac s law avogadros law and molar volume at stp combined gas law problems. If it is released. Some of the worksheets below are combined gas law problems worksheet answer key gas laws worksheet.
The volume changes from 45 l to 40 l.

Gas Law Quiz Dalton S Law Graham S Law Ideal Gas Law Ideal Gas Law Dalton S Law Science Chemistry

Gas Laws Crossword Puzzle And Answer Key This Crossword Puzzle Contains Vocabulary Per Teaching Middle School Science High School Chemistry High School Science

Boyle 039 S Law Worksheet Answer Key Elegant Boyle S Law Worksheets Answers In 2020 Nouns Worksheet Worksheets Possessive Nouns Worksheets

42 Math Conversion Worksheets For Grade 7 Di 2020

Gas Laws Bundle Practice Problem Worksheets Homework Worksheets Teaching Chemistry Chemistry Education

Chemistry Gas Laws Matching Puzzle Ideal Gas Law Chemistry Study Guide Gas Laws Chemistry

Lines Combined Gas Law Worksheet Chart Answer Key Check More At Https Oakleys Sunglasses Top Bobby Black Combined Ideal Gas Law Worksheets Chemical Equation

Pin By Crissy Jane Mirasol On Math In 2020 Chemistry Notes Physics And Mathematics Chemistry Lessons

Pin On Creations By Callie Teacher Pay Teachers

Gas Laws Editable Powerpoint Examples Included Distance Learning In 2020 Ideal Gas Law Dalton S Law Distance Learning

Pin On Creations By Callie Teacher Pay Teachers

Comparing The Gas Laws Table Literal Equations Word Problem Worksheets Gas Laws Chemistry

Charles Law Chemistry Notes Gas Laws Chemistry Chemistry

Combined Gas Law Practical Application Ideal Gas Law Grammar Worksheets Ideal Gas Law Gas Laws Chemistry

Boyle039s Law And Charles Law Gizmo Worksheet Answers Ideal Gas Law Charles Law Gas Laws Chemistry

Chemistry Gas Laws Boyle S Law Chemistry Lessons Science Teaching Resources Chemistry





