Balance the following equations and indicate the type of reaction as formation decomposition single replacement double replacement hydrocarbon combustion or other. 4k 2o 2 2k 2 o 29.

Balancing Chemical Equations Worksheets Combo In 2020 Chemical Equation Balancing Equations Equations
Fe h 2s0 4.

Chemical equations worksheet. Chemical equations are balanced for mass and charge meaning the number and type of atoms on the left side of the arrow is the same as the number of type of atoms on the. Balancing chemical equations worksheet 2 answers 26. Identify the parts of a chemical equation describing a chemical reaction using words and symbolic equations with several exercise and answers.
2mg cl 2 mgcl 2 27. Cl 2 3f 2 2clf 3 30. K 2co 3 hno 3.
A balanced chemical equation is useful in showing us the ratios of reactants and products. Every balanced chemical equation consists of two parts. Nh 3 o 2.
The equation identifies the reactants starting materials and products resulting substances the formulas of the participants the phases of the participants solid liquid gas the direction of the chemical reaction and the amount of each substance. 2ag 2 o 4ag o 2 28. Both of these sides are separated by the means of an arrow.
B no 3 3 hbr. Nh 4 3po 4 pb no 3 4. Sio 2 2c si 2co 31.
Sn h 2o. The reactant side and the product side. Writing and balancing equations worksheet.
Sno 2 h 2. B 2 so 3 3 lif. Cu s o2 g cuo s b.
C 2h 6 o 2. 2zns 3o 2 2zno 2so 2 33. A chemical equation describes what happens in a chemical reaction.
The amount of reactants must be equal to the amount of products. Kno 3 h 2co 3. Displaying top 8 worksheets found for balancing chemical equations answer sheet.
The chemical equation has the products on the right side while the reactants are written on the left side. Balancing an unbalanced equation is mostly a matter of making certain mass and charge are balanced on the reactants and products side of the reaction arrow. B 2br 6 hno 3.
No h 2o. Writing and balancing chemical reactions 1. Chemical equation a chemical equation shows the reactants and the products of a chemical reaction.
This side represents the elements which are used for initiating the experiment. 4fe 3o 2 2fe 2 o 3 36. For instance 2h2 o2 2h20 denotes that there are four atoms of hydrogen and 2 atoms of oxygen on both sides of the equation.
So 2 2h 2 s 3s 2h 2 o 34. 3 bf li 2so 3. 2nahco 3 na 2 co 3 co 2 h 2 o 32.
H 2o co 2. Both of them are separated by an arrow. Balancing chemical equations is necessary because the same atoms should be present before and after the reaction just in different arrangements.
K 3po 4 h 2o. 2ba o 2 2bao 35. Koh h 3po 4.
Some of the worksheets for this concept are balancing equations practice problems teacher answer balancing equations balancing chemical equations answer chemical formulas equations work answers another balancing equation answer key balancing equations work answers balancing word equations chapter 9. A balanced chemical equation gives the number and type of atoms participating in a reaction the reactants products and direction of the reaction. Balance the following chemical equations.
Fe 2 so 4 3 h 2. On the left side of the arrow you will find the reactant side.

Download Balancing Equations 07 Balancing Equations Chemical Equation Literal Equations

Download Balancing Equations 08 Balancing Equations Chemistry Worksheets Chemical Equation

Download Balancing Equations 09 Balancing Equations Balancing Equations Chemistry Chemical Equation

Balancing Equations Worksheet Moercar Balancing Equations Chemical Equation Chemistry Worksheets

Balancing Equations 06 Balancing Equations Practices Worksheets Chemical Equation

Download Balancing Equations 14 Chemical Equation Equations Balancing Equations

Image Result For Balancing Chemical Equations Worksheet Balancing Equations Chemical Equation Chemistry Worksheets

Balancing Chemical Equations Chemical Equation Balancing Equations Chemistry Worksheets

Balancing Chemical Equations Worksheets With Answers Chemical Equation Balancing Equations Chemistry Worksheets

Balancing Chemical Equations Chemical Equation Chemistry Worksheets Balancing Equations

Balancing Chemical Equations Worksheet Chemistry Education Chemistry Lessons Chemistry Classroom

Printable Sample Balancing Equations Worksheet Form Chemical Equation Balancing Equations Equations

Download Balancing Equations 26 Balancing Equations Literal Equations Chemical Equation

Balancing Equations Worksheet Stem Sheets Chemistry Worksheets Chemical Equation Balancing Equations
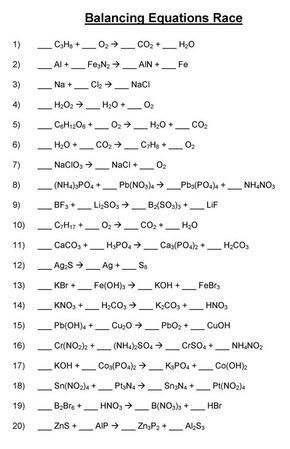
Writing And Balancing Chemical Equations Worksheet In 2020 Chemistry Lessons Teaching Chemistry Chemistry Education

Balancing Chemical Equations Practice Worksheet With Answers Chemical Equation Practices Worksheets Balancing Equations Chemistry

Balancing Chemical Equations Worksheets With Answers Chemical Equation Balancing Equations Equations

Do You Find Balancing The Chemical Equation A Daunting Task Download Our Balancing Chemical Equations Workshee Balancing Equations Chemical Equation Equations

Balancing Equations Worksheet Chemical Equation Dimensional Analysis Chemistry Worksheets


