13 12 Here are three isotopes of an element. Isotope and Ions Practice Worksheet.
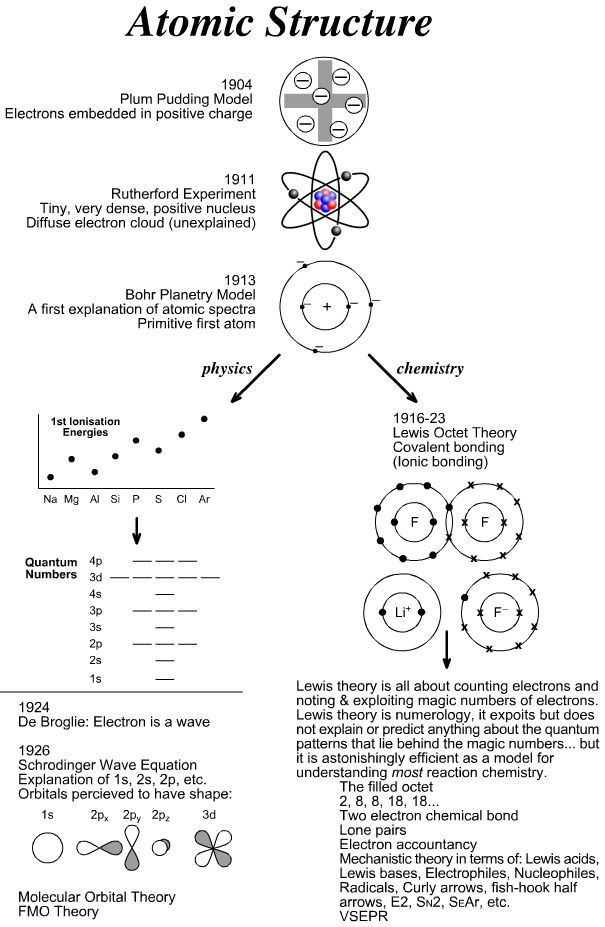
Timeline Structural Theory Chemogenesis Chemistry Chemistry Lessons Teaching Chemistry
Subatomic particle counting worksheet answers.

Atoms and ions worksheet answer key. The number 6 refers to the c. Mole Calculation Worksheet Answer Key 1 How many moles are in 15 grams of lithium. The buildup of hydrogen ions makes the stroma negatively charged relative to the space within the thy-lakoids.
These worksheet also relieve any worries a teachers may have for students that miss class for whatever reason. All atoms are neutral. And indicate the proportionate number of atoms of each element in this chemical formulas worksheet pdf.
It is that simple. When energy is added to a gas its atoms get very excited and become ions when the outer electrons of atoms break off creating plasma. Atomfion of an element has the same number of lostgained electrons.
Based on this information list the requirements for two atoms to be isotopes of each other. How many protons and neutrons are in the first isotope. Then indicate what type of bonding is holding the atoms together in one molecule of the following.
Explain your answer fully or- 10 ses Label the parts of an atom on the diagram below. Consider the ions of potassium K and sulfur S. D4C-e–vnvt Test your understanding of isotopes by examining the relationships between the pairs of atoms listed below.
Some of the worksheets for this concept are Particle theory work Subatomic particle counting work answers Particle model of light work 1a answers. Five Types of Chemical Reaction Worksheet Answers. B An Q tom protons as electrons.
However we do find exceptions to the order of filling of orbitals that are shown in Figure 3 or Figure 4For instance the electron configurations shown in Figure 6 of the transition metals chromium Cr. Use the ions given above to work out the formulae of the following ionic compounds. Element Name Atomic Number Mass Number Number of protons Number of neutrons Number of electrons Isotopic notation oxygen 8 17 8 9 8 17 😯 phosphorous 15 31 15 16 15 31 15 P.
Are located in the nucleus of an atom. An awesome collection of free atomic structure worksheets for teachers. 181 10 S atoms 1 mole S 602 x 10 S atoms 1 3 moles S 24 23 u u.
The amount of reactants must be equal to the amount of products. Consider the ions of iron Fe and sulfur S. IMF Intermolecular Forces Worksheet Indicate the strongest IMF holding together thousands of molecules of the following.
The numbers 12 13 and 14 refer to the d. Paste each image into a blank document to turn in with this worksheet. When students get big chemical equations in a balancing equation worksheet they often find it to be very difficult.
This is a modified version of the activity created by Timoty Herzog and Emily Moor. How many atoms in 3 moles of sulfur. Keywords atoms ions isotopes.
The of an element is the average mass of an elements naturally occurring atoms or isotopes taking into account the of each isotope. By doing this atoms form bonds. Mass Number Number of Protons Number of Electrons Number of Neutrons 23 39 19 38 38 50 20 40 Ions 2 -1 Isoto es 110 47 36S.
Write chemical formulas for all possible ionic compounds involving these ions using the simplest ratios of potassium K and sulfur S. Complete the following sentences using the terms in parentheses. An acid and a.
Keep in mind that the sum of the charges in an ionic compound must equal zero. Carbon-14 of protons 6 6 6 of neutrons 6 7 8 of electrons 6 6 6 Chromium-58 Chromium-63 of protons 24 24 of neutrons 34 39 of electrons 24 24 Nitrogen-15 Nitrogen-20. An atom is the smallest constituent unit of ordinary matter.
Level High School Middle School. 4 C 5 H 9 O 29 O 2 20 CO 2 18 H 2 O combustion 4. Electrons in the correct orbitals and to fill out the key for the subatomic.
Hydrogen ions began to accumulate within the thylakoid space. This gradient the difference in both charge and H ion concentration across the membrane provides the energy to make ATP. The periodic table can be a powerful tool in predicting the electron configuration of an element.
Isotope Practice Worksheet Name. Write chemical formulas for all possible ionic compounds involving these ions using the simplest ratios of potassium K and sulfur S. Assume all atoms are neutral.
Because of the arrangement of the atoms and electrons in a water molecule there are two differently charged areas of. What is the charge of an ion. It is composed of protons neutrons and electrons.
4 Some Types of Chemical Reactions Although there are untold millions of. For instance 2H2 O2 – 2H20 denotes that there are four atoms of hydrogen and 2 atoms of oxygen on both sides of the equation. ATP synthase allows H ions to pass through the.
What is the charge of an atom. Atoms of the same element with the same Z C. Take the hassle out of reviewing with this pdf to tell the difference between key terms like atoms and molecules elements and compounds homogeneous and heterogeneous mixtures and more.
The use of these worksheets also encourages their reading skills. Atomic number 29 among others are not those we would. Pdf Ionic Equation Activity ANSWER KEY.
The students are instructed to bond with other ions and keep a record of their bonds. Com Virtual labs are safer for students to practice in than a real lab if the students are unfamiliar with the safety protocol. The students fill in the Atoms Family worksheet as we discuss the presentation and then practice singing the song.
The atoms in the previous question are isotopes of each other. Atomic Structure Worksheet What type of charge does a proton have. 1 mole sulfur 602 x 1023 S atoms Answer.
Hand them the worksheet they missed and be done with it. Write the ionic charges such as Ca 2. The bonding electrons in some molecules are not equally shared between the atoms.
– Periodic Table Basics 1 pdf includes worksheet answer key and Periodic Table Basics 1 cards pdf Sample cards. NOTE if the molecule is an ionic compound then there is no IMF the ions are all held together by ionic bonds. Answer key is included.
Atomic number 24 and copper Cu. Keep in mind that the sum of the charges in an ionic compound must equal zero. For each of the following isotopes write the number of protons neutrons and electrons.
The subatomic particle with no electrical charge is the 2. Consider the ions of potassium K and sulfur S. Get more information about atoms on our website.
These neutral molecules with a difference of charge across the molecule are called polar molecules. SUBATOMIC PARTICLES and ISOTOPES WORKSHEET Complete the following table using the information discussed in class and your Periodic Table. The number game with atoms and ions 1.
Atoms are the smallest pieces of all matter. Atom I 12 Carbon- 12 Argon-40 11 An atom with 13 protons and 13 neutrons. If a substance is neutral it has an equal amount of positive and negative ions present in it.
Just like students sharing markers atoms sometimes share or swap electrons. If a liquid has a greater concentration of positive ions in the form of hydrogen than say water it is classified as an acid. If the liquid has less hydrogen ions than water it is a base.
A The atomic numbermass of an element is the same as the number of protons in the nucleus of an atom. ANSWER KEY June 04 2019. How many protons and neutrons are in the second isotope.

Chem Unit 2 Worksheet Bundle Atoms Ions Isotopes Compo Chemistry Worksheets Teaching Chemistry Scientific Method Worksheet

Atoms And Ions Worksheet New Isotopes Worksheet Pdf Answers Chemistry Worksheets Worksheets Worksheet Template

Proton Neutron Electron Scientist Practice Electron Configuration Electrons Protons

Ionic Bonds Worksheet Chemistry Worksheets Ionic Bonding Covalent Bonding

Graphing Periodic Trends Activity Graphing Worksheets Graphing Chemistry Worksheets

Lewis Dot Structure Mini Lesson And Worksheet Chemistry Worksheets Mini Lessons Teaching Chemistry

Practice Determining The Number Of Protons Neutrons And Electrons With This Clever Chemistry Review Chemistry Worksheets Chemistry Classroom Science Education

Protons Neutrons And Electrons Practice Worksheet Practices Worksheets Text Features Worksheet Chemistry Worksheets

Electronic Shell Worksheet Electron Sheath Chart Science Education Teaching Chemistry Chemistry Worksheets Chemistry Classroom

Warmup Quiz Mini Assignment Ionic Charges Cation Anion Bonding Practice Middle School Science Resources Chemistry Worksheets Graphing Quadratics

Bohr Model Worksheet Answers Fresh Drawing Bohr Models Worksheet Chemistry Worksheets Bohr Model Atomic Structure

Electron And Molecular Geometries Molecular Geometry Chemistry Teaching Chemistry

Atoms Molecules Ions Naming Ionic Compounds Chemistry Notes Teaching Chemistry Study Chemistry

Worksheet Can Be Used As Homework Or An Assessment Covers Elements Atomic Mass Isotopes Fraction Word Problems Chemistry Worksheets Solving Linear Equations

Counting Atoms Worksheet Editable Printable Distance Learning Options Counting Atoms Counting Atoms Worksheet Chemistry Worksheets

Ions And Isotopes Practice Two Versions Practices Worksheets Multi Step Equations Worksheets Fraction Word Problems

Bohr Model Worksheet Answers Tecnologialinstante Bohr Model Chemistry Worksheets Chemistry

Atom Element Molecule Or Compound Teaching Chemistry Chemistry Classroom Matter Science

Modeling Isotopes And Ions Science Chemistry Science Lessons Physical Science

