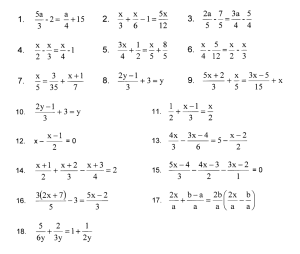In other words stoichiometry is the practice of using a chemical reaction equation to predict the results of the. Worksheet solutions by unit.

Ap Chemistry Master Cheatsheet Ap Chemistry Chemistry Lessons College Notes
Ap stoichiometry 1.

Ap chemistry solution stoichiometry worksheet. Stoichiometry derives from the greek stoicheionmetron meaning element measure. Taking full advantage of all features presented in peterson s master ap chemistry you will become much more comfortable with the test and considerably more confident about getting a high score. Ap chemistry chapter 4.
Stoichiometry multiple choice answers 44. Empirical formula and percent composition. What number of moles of o 2 is needed to produce 14 2 grams of p 4o 10 from p.
Calculate the molarity of each of the following solutions. Unit 3 stoichiometry and equations worksheet 1 memorize. A 16 45 g sample of nacl is dissolved in enough water to make 1 000 l of solution b.
How many grams of silver chromate will precipitate when 150. Aqueous reactions and solution stoichiometry 3 4 2 precipitation reactions reactions that result in the formation of an insoluble product are known as precipitation reactions. Solution stoichiometry deals with all types of solutions but mostly with aqueous ones because that is where we live and function.
Content resources bozemanscience chemicool ck12 chem 2nd ed crashcourseapchem. Solution stoichiometry worksheet solve the following solutions stoichiometry problems. Appendix peterson s college by college guide to ap credit and placement gives you the equivalent.
Molar mass p 4o 10 284 a 0 0500 mole b 0 0625 mole c 0 125 mole d 0 250 mole e 0 500 mole 4 p 5 o. Ml of 0 400 m potassium chromate. Ml of 0 500 m silver nitrate are added to 100.
Ap chemistry michalek unit 4a solution stoichiometry 7 worksheet 4b 01 name molarity 1. Worksheet 4 heat of solution write up. Stoichiometry is one of the most important topics on the ap chemistry exam so it s vital that you understand it and all of its applications.
Name chemistry 110. Solution chemistry ap question worksheet pdf 93k valerie brewer sep 21 2016 4 11 pm. 1 how many grams of calcium phosphate can be produced from the reaction of 2 50 l of 0 250 m calcium chloride with and excess of phosphoric acid.
A precipitate is an insoluble solid formed by a reaction in solution. 2 agno 3 aq k 2 cro 4 aq ag 2 cro 4 s 2 kno 3 aq 0 150 l agno 3 0 500 moles agno 3 1 moles ag 2 cro 4 331. A 853 0 mg sample of kio 3.

Stoichiometry Map 2 Worksheets Bundle Great Learning Tool Chemistry Chemistry Worksheets Chemistry Lessons Chemistry Basics

Skillbuilder 8 5 Unit 1 Stoichiometry Kids Worksheets Printables Cursive Writing Worksheets Stoichiometry Chemistry

This Set Of Stoichiometry Worksheets Covers Empirical Formulas Molecular Formulas Percent Compos Chemistry Worksheets Chemistry Education Chemistry Classroom

Chemistry Notes Chemistry Pdf Chemical Kinetics And Rate Laws Chemical Kinetics Chemistry Notes Engineering Notes

Properties Of Solutions In 2020 Chemistry Notes Chemistry Education Study Chemistry

Stoichiometry Particle Diagram Chemistry Worksheets Apologia Chemistry Teaching Chemistry

Chemistry Notes Types Of Chemical Reactions Solution Stoichiometry In 2020 Chemistry Notes Chemical Reactions Chemistry

Skillbuilder 8 3 Unit 1 Stoichiometry Chemistry The Unit Book Lovers

4 4 Solution Concentration And Solution Stoichiometry Solutions Chemistry Bottle

Chemistry Notes Types Of Chemical Reactions Solution Stoichiometry In 2020 Chemistry Notes Equations Notes Chemistry

Stoichiometry Problems Answers 4 Chemistry Worksheets College Chemistry Chemistry Lessons

The Chemistry Close Read Chemistry Solutions Vol 2 Issue 1 Chemistry Education Chemistry Activities Chemistry Classroom

Chemistry Mole Concept Worksheet In 2020 Mole Concept Chemistry Preschool Learning Activities

Seven Stoichiometry Worksheets With Keys Worksheets Staar Teaching

Unit 2 2016 Solution Stoich Mr Klamm Ap Chemistry Ap Chemistry Chemistry The Unit

Stoichiometry And Avogadro S Principle Gas Stoichiometry Worksheet Kids Worksheets Printables Chemistry Lessons Worksheets

Skillbuilder 8 6 Chemistry Worksheets Chemistry Education Chemistry

Organic Chemistry Formulas Chemistry In 2020 Organic Chemistry Notes Chemistry Notes Organic Chemistry Study